- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 12:10 | Herantis Pharma plc: Herantis reports positive FDA feedback and finalizes Phase 2a study design for HER-096 | 205 | GlobeNewswire (Europe) | FDA considered the planned HER-096 Phase 2a study design appropriateStudy design finalized; approximately 100 patients to be enrolledMore than 50% of Phase 2 funding secured or identified
Espoo... ► Artikel lesen | |
| 11:20 | Herantis Pharma Oyj: Herantis reports positive FDA feedback and finalizes Phase 2a study design for HER-096 | 35 | GlobeNewswire (Europe) | Herantis Pharma Plc - Press release on 2 June 2026 at 12:20 EEST
Herantis reports positive FDA feedback and finalizes Phase 2a study design for HER-096
FDA considered the planned HER-096 Phase 2a study... ► Artikel lesen | |
| 19.05. | Herantis Pharma Oyj: Herantis Pharma partners with Indivi to enable detection of early treatment effects in upcoming HER-096 Phase 2 trial in Parkinson's disease | 164 | GlobeNewswire (Europe) | Herantis Pharma Plc - Press release 19 May 2026 at 09:00 EEST
Herantis Pharma partners with Indivi to enable detection of early treatment effects in upcoming HER-096 Phase 2 trial in Parkinson's disease... ► Artikel lesen | |
| 12.05. | Herantis Pharma Oyj: Herantis Pharma Plc's board decides on new option rights program for managing director, management team and other key personnel | 95 | GlobeNewswire (Europe) | Herantis Pharma PlcCompany release, 12 May 2026 at 19:15 EEST
Herantis Pharma Plc's board decides on new option rights program for managing director, management team and other key personnel
The Board... ► Artikel lesen | |
| 23.04. | Herantis Pharma Oyj: Decisions of Herantis Pharma Plc's Annual General Meeting of shareholders and constitutive meeting of the Board of Directors | 170 | GlobeNewswire (Europe) | Herantis Pharma Plc
Company release, 23 April 2026 at 4:50 p.m. Eastern European Summer Time
Decisions of Herantis Pharma Plc's Annual General Meeting of shareholders and constitutive meeting of the... ► Artikel lesen | |
| 16.03. | Herantis präsentiert positive Studiendaten zu Parkinson-Medikament auf Fachkonferenz | 2 | Investing.com Deutsch | ||
| 09.03. | Herantis Pharma - Momentum continues into FY26 | 361 | Edison Investment Research | Herantis Pharma's FY25 results reflect positive momentum for HER-096, which is being developed as a potential disease-modifier for Parkinson's disease (PD). Notably, the candidate successfully completed... ► Artikel lesen | |
| HERANTIS PHARMA Aktie jetzt für 0€ handeln | |||||
| 05.03. | Herantis Pharma: Stabile Finanzen und Studienerfolg - Kapital für Phase-2-Studie gesucht | 2 | Investing.com Deutsch | ||
| 05.03. | Herantis Pharma Oyj: Herantis Pharma releases 2H and FY 2025 report today | 288 | GlobeNewswire (Europe) | Company announcement
Herantis Pharma Plc | Company Release | March 5, 2026 at 08:00 EET
Herantis Pharma releases 2H and FY 2025 report today
Espoo, Finland, March 5, 2026: Herantis Pharma Plc ("Herantis")... ► Artikel lesen | |
| 02.03. | Herantis Pharma Oyj: Directed Issue | 191 | GlobeNewswire | NOTICE, MARCH 02, 2026 SHARES (Record Id 336048) A total of 571,429 shares will be traded as old shares as of March 03, 2026 on First North Growth Market... ► Artikel lesen | |
| 20.02. | Herantis Pharma - Funding to support HER-096 in Phase II | 595 | Edison Investment Research | Herantis Pharma has made tangible progress towards raising funds to support its Phase II proof-of-concept study for lead asset HER-096 as a potential disease-modifier for Parkinson's disease (PD). In... ► Artikel lesen | |
| 19.02. | Herantis Pharma plc: Inside information: Herantis Pharma selected for Horizon 2025 grant of EUR 8.0 million for Phase 2 trial of HER-096 | 521 | GlobeNewswire (Europe) | Funding to support the execution of a Phase 2 proof-of-concept trial of HER-096 in Parkinson's disease, following completion of study preparations
Espoo, Finland, 19 February 2026: Herantis Pharma... ► Artikel lesen | |
| 11.02. | Herantis Pharma Oyj: Inside information: Herantis Pharma Plc successfully completes a directed share issue raising EUR 4.2 million | 231 | GlobeNewswire (Europe) | Herantis Pharma PlcCompany release, inside information, 11 February 2026 at 23:45 (EET)
Inside information: Herantis Pharma Plc successfully completes a directed share issue raising EUR 4.2 million
THE... ► Artikel lesen | |
| 08.01. | Herantis Pharma - Biomarker data bolster HER-096 potential | 500 | Edison Investment Research | Herantis Pharma has reported encouraging results from the biomarker component of its Phase Ib trial testing HER-096 in patients with Parkinson's disease (PD). The key takeaway was that the data showed... ► Artikel lesen | |
| 07.01. | Herantis Pharma reports positive biomarker data for Parkinson's drug | 1 | Investing.com | ||
| 07.01. | Positive Biomarker-Daten zu Parkinson-Wirkstoff beflügeln Herantis-Pharma-Aktie | 9 | Investing.com Deutsch | ||
| 07.01. | Herantis Pharma plc: Herantis Pharma - Phase 1b biomarker data show clear evidence of biological response to HER-096 in people with Parkinson's disease | 491 | GlobeNewswire (Europe) | Herantis Pharma Plc - Company release
Inside information: Herantis Pharma - Phase 1b biomarker data show clear evidence of biological response to HER-096 in people with Parkinson's disease
Biomarker... ► Artikel lesen | |
| 26.11.25 | Herantis Pharma plc: Herantis Pharma announces successful completion of six-month GLP toxicology study of HER-096 | 258 | GlobeNewswire (Europe) | Key step achieved in advancing HER-096 towards Phase 2 efficacy trial Espoo, Finland, 26 November 2025: Herantis Pharma Plc ("Herantis"), a clinical-stage company developing disease-modifying therapies... ► Artikel lesen | |
| 08.10.25 | Herantis Pharma - HER-096 clears Phase Ib, eyes Phase II | 521 | Edison Investment Research | Herantis Pharma reported positive top-line results from the second part of its Phase Ib trial, evaluating lead candidate HER-096 in Parkinson's disease (PD) patients, meeting both the primary and secondary... ► Artikel lesen | |
| 07.10.25 | Herantis Pharma plc: Herantis Pharma presents positive topline data for HER-096 in Phase 1b trial for people living with Parkinson's disease | 535 | GlobeNewswire (Europe) | Herantis Pharma announces positive topline data for HER-096 in Phase 1b trial for people living with Parkinson's disease
Trial meets all primary and secondary endpoints establishing a strong... ► Artikel lesen |
1 2 Weiter >>
24 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| NOVO NORDISK | 37,750 | -1,05 % | Novo Nordisk-Aktie: Droht jetzt ein Kurssturz um -30%? | Der jüngste Aufwärtstrend der Novo Nordisk-Aktie ging Mitte Mai zu Ende. Der Kurs des dänischen Pharmakonzerns schaffte es nicht mehr über die Hürde von 300 DKK. Und nun droht nach Einschätzung eines... ► Artikel lesen | |
| MERCK KGAA | 130,80 | 0,00 % | DAX-Check LIVE: Delivery Hero, Infineon, Merck, Nordex, Secunet, Siemens im Fokus | Der deutsche Aktienmarkt hat den Pfingstmontag mit einem fulminanten Kurssprung begangen. Der DAX startete mit Gewinnen, übersprang direkt die wichtige Marke von 25.000 Punkten und kletterte im Verlauf... ► Artikel lesen | |
| AURORA CANNABIS | 3,144 | +0,58 % | War das erst der Anfang? Warum der Aurora-Deal und ein 50-Millionen-AUD-Vertrag Bioxyne weiteren Schub geben könnten | ||
| GSK | 21,170 | -0,38 % | GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff | DJ GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff
Von Adria Calatayud
DOW JONES--GSK hat mit einem Medikamentenkandidaten gegen Hepatitis B einen teilweisen Therapieerfolg... ► Artikel lesen | |
| ABBVIE | 183,30 | +0,11 % | AbbVie Announces European Commission Approval of AQUIPTA (atogepant) for the Acute Treatment of Migraine in Adults | AQUIPTA (atogepant) is now approved in the European Union for the acute treatment of migraine in adults, with or without aura, and for the prophylaxis of migraine in adults who have at least... ► Artikel lesen | |
| ROCHE | 347,70 | -0,18 % | BARCLAYS stuft ROCHE HOLDINGS AG auf 'Overweight' | LONDON (dpa-AFX Analyser) - Die britische Investmentbank Barclays hat die Einstufung für Roche mit einem Kursziel von 410 Franken auf "Overweight" belassen. Analyst James Gordon berichtete am Montagabend... ► Artikel lesen | |
| CANOPY GROWTH | 0,935 | +0,45 % | Canopy Growth Corporation: Canopy Growth Relaunches Tweed Brand in Germany with New MTL Cannabis Strain Lineup, Marking First International Release following Acquisition | Pablo's Revenge, Dante'z Inferno, and Frost'd Flakes launched under Tweed brand Up to five additional strains expected to follow in June 2026 Company targeting rapid growth in Germany's medical... ► Artikel lesen | |
| STADA ARZNEIMITTEL | - | - | Enzymatica AB: Quarterly report Q1/2026 - Enzymatica strengthens international position through STADA agreement | First quarter
Net sales amounted to SEK 11.4 million (12.3).
Operating result amounted to SEK -13.6.million (-18.0)
Earnings per share, before... ► Artikel lesen | |
| ELI LILLY | 929,40 | -0,10 % | HBM Healthcare Investments AG: Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden | HBM Healthcare Investments AG
/ Schlagwort(e): Ankauf
Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden
26.05.2026 / 13:10 CET/CEST
Die... ► Artikel lesen | |
| ASTRAZENECA | 155,00 | +0,32 % | Astrazeneca liefert FDA neue Daten für US-Zulassung von Camizestrant nach | DJ Astrazeneca liefert FDA neue Daten für US-Zulassung von Camizestrant nach
Von Adria Calatayud
DOW JONES--Die US-Arzneimittelbehörde FDA verschiebt ihre Entscheidung über die Zulassung eines... ► Artikel lesen | |
| BRISTOL-MYERS SQUIBB | 47,310 | +0,17 % | BioNTech SE: Globale Daten der ROSETTA Lung-02-Studie: Ermutigende Wirksamkeit für BioNTechs und Bristol Myers Squibbs bispezifischen PD-L1xVEGF-A-Immunmodulator-Kandidat Pumitamig bei nicht-kleinzelligem Lungenkrebs | Erster bispezifischer PD-(L)1xVEGF-Immunmodulator-Kandidat, zu dem globale Daten mit ermutigender Wirksamkeit in Kombination mit Chemotherapie in der Erstlinienbehandlung von nicht-kleinzelligem Lungenkrebs... ► Artikel lesen | |
| TEVA | 29,800 | 0,00 % | Teva Pharmaceutical Industries Ltd: Teva to Present at the Goldman Sachs Global Healthcare Conference | PARSIPPANY, N.J., and TEL AVIV, Israel, June 01, 2026 (GLOBE NEWSWIRE) -- Teva Pharmaceutical Industries Ltd. (NYSE and TASE: TEVA) today announced that Richard Francis, Teva's President and CEO, will... ► Artikel lesen | |
| VERTEX PHARMACEUTICALS | 375,95 | -0,27 % | Vertex: FDA Accepts BLA Submission For Povetacicept; Assigns PDUFA Target Action Date Of Nov. 30 | CAMBRIDGE (MASSACHUSETTS) (dpa-AFX) - Vertex Pharmaceuticals Incorporated (VRTX) announced the FDA has accepted its Biologics License Application submission for povetacicept, an investigational... ► Artikel lesen | |
| BIOTEST | 32,400 | -1,52 % | Biotest erhält FDA-Zulassung | Summary: FDA lässt Fibrinogen-Produkt für den US-Markt zu Biotest entwickelt und produziert, Grifols vermarktet Markteinführung für 2026 geplant Pipeline gewinnt strategisch an Gewicht Biotest hat einen... ► Artikel lesen | |
| ZOETIS | 66,74 | +0,09 % | Argus downgrades Zoetis stock rating on competition, margin pressure |
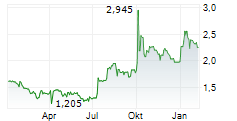
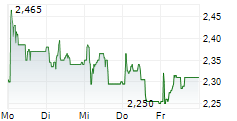
HERANTIS PHARMA OYJ gehört der Branche Pharma an. Mit einer aktuellen Kursveränderung von +0,043 (+1,90 %) liegt der Kurs bei 2,283 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu HERANTIS PHARMA OYJ finden Sie auf Wallstreet Online.