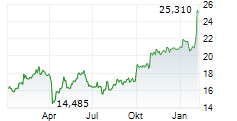
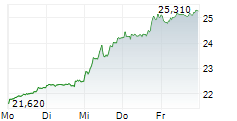
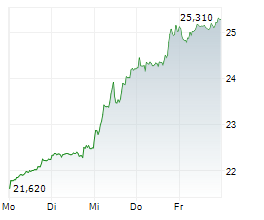
LONDON (dpa-AFX) - GSK plc (GSK) on Thursday said Japan's Ministry of Health, Labour and Welfare has accepted a new drug application for bepirovirsen for the treatment of adults with chronic hepatitis B (CHB).
The submission is supported by positive results from the Phase III B-Well 1 and B-Well 2 trials, in which bepirovirsen demonstrated a statistically significant and clinically meaningful functional cure rate. The drug candidate also showed an acceptable safety and tolerability profile consistent with previous studies.
Chronic hepatitis B affects more than 250 million people globally and nearly one million people in Japan.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News