NEW YORK CITY (dpa-AFX) - Pfizer Inc. (PFE) and Astellas Pharma Inc., Friday announced the positive results from the Phase 3 EV-304 clinical trial for PADCEV in combination with Keytruda in patients with muscle-invasive bladder cancer.
Before and after surgery data revealed a 47% reduction in the risk of tumor recurrence, progression or death compared to patients treated with standard of care neoadjuvant gemcitabine and cisplatin.
Additionally, an estimated 79.4% of patients were event-free at two years, relative to 66.2% treated with standard of care neoadjuvant chemotherapy.
Further, the study showed a 35% reduced risk of death in patients treated with perioperative enfortumab vedotin plus pembrolizumab versus neoadjuvant chemotherapy.
The EV-304 results, combined with the EV-303 study, provide compelling evidence that perioperative enfortumab vedotin plus pembrolizumab may offer survival benefits in the curative setting for patients with muscle-invasive bladder cancer, the company stated.
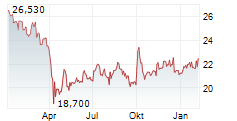
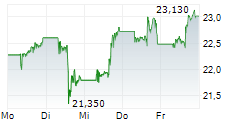
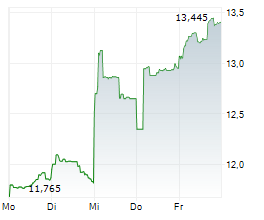
Currently, PFE is trading at $27.28, up 0.64 percent on the New York Stock Exchange.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News