WASHINGTON (dpa-AFX) - BioMarin Pharmaceutical Inc. (BMRN) announced Friday that the U.S. Food and Drug Administration has approved the company's supplemental Biologics License Application for PALYNZIQ (pegvaliase-pqpz) to include pediatric patients 12 years of age and older with phenylketonuria.
PALYNZIQ is the only enzyme substitution therapy approved to reduce blood phenylalanine (Phe) concentrations in people with phenylketonuria.
The company said it is also seeking approval from the European Medicines Agency with the goal of expanding treatment with PALYNZIQ to include adolescents as young as age 12 in the European Union.
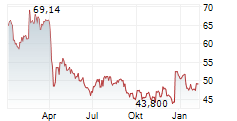
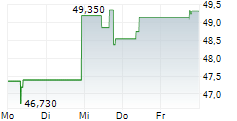
BMRN closed Friday's regular trading at $61.73, up $0.62 or 1.01%. However, in after-hours trading, the stock slipped to $59.31, down $2.42 or 3.92%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News