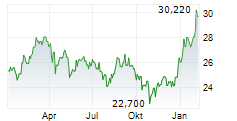
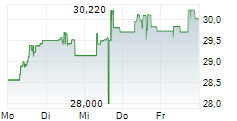
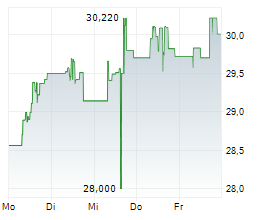
TOKYO (dpa-AFX) - Takeda Pharmaceutical Company Limited (TAK) and Protagonist Therapeutics, Inc. (PTGX) said Monday the U.S. Food and Drug Administration has accepted the New Drug Application for rusfertide and granted Priority Review for the treatment of adults with polycythemia vera.
The FDA has set a Prescription Drug User Fee Act goal date in the third quarter of this year.
Polycythemia vera is a rare blood disorder marked by overproduction of red blood cells, which thickens the blood and increases the risk of life-threatening blood clots.
The application is based primarily on positive 32-week primary analysis and 52-week results from the Phase 3 VERIFY study, along with four-year data from the Phase 2 REVIVE study and long-term extension THRIVE study.
Under a 2024 global licensing and collaboration agreement, Protagonist Therapeutics, which discovered rusfertide, has the option to co-commercialize the drug in the U.S. under a 50/50 profit-and-loss share or opt out and grant Takeda a worldwide license.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News