| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 27,050 | 27,360 | 11:20 | |
| 27,050 | 27,320 | 11:20 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Di | TAKEDA PHARMACEUTICAL COMPANY LIMITED: GENERAL ANNOUNCEMENT::NOTICE CONCERNING DISPOSAL OF TREASURY SHARES - LTIP FOR COMPANY GROUP EMPLOYEE OUTSIDE OF JAPAN | 9 | SGX Company Announcements | ||
| Mo | Incyte inks $2B deal to add Star unit to late-phase constellation and challenge Takeda | 13 | FierceBiotech | ||
| Fr | Takeda takes $2.5B legal charge after pay-for-delay verdict, swings to loss for '25 | 21 | FiercePharma | ||
| Fr | TAKEDA PHARMACEUTICAL CO LTD - 6-K, Report of foreign issuer | 1 | SEC Filings | ||
| Fr | TAKEDA PHARMACEUTICAL COMPANY LIMITED: GENERAL ANNOUNCEMENT::REVISION OF FY2025 CONSOLIDATED FINANCIAL STATEMENTS | 15 | SGX Company Announcements | ||
| TAKEDA PHARMACEUTICAL Aktie jetzt für 0€ handeln | |||||
| Fr | TAKEDA PHARMACEUTICAL COMPANY LIMITED: FINANCIAL STATEMENTS AND RELATED ANNOUNCEMENT::FULL YEARLY RESULTS | 20 | SGX Company Announcements | ||
| Do | Innovent, fresh from $1.2B deal, posts phase 3 win for Takeda-partnered ADC | 13 | FierceBiotech | ||
| 01.06. | Berlin: FÜRST gewinnt Takeda Pharma als neuen Büromieter | 16 | DEAL-Magazin | ||
| 01.06. | TAKEDA PHARMACEUTICAL COMPANY LIMITED: GENERAL ANNOUNCEMENT::WITHDRAWAL OF SHELF REGISTRATION STATEMENT AND FILING OF NEW SHELF REGISTRATION STATEMENT IN JAPAN | 11 | SGX Company Announcements | ||
| 26.05. | TAKEDA PHARMACEUTICAL CO LTD - 6-K, Report of foreign issuer | 13 | SEC Filings | ||
| 26.05. | TAKEDA PHARMACEUTICAL COMPANY LIMITED: GENERAL ANNOUNCEMENT::PLANNED REVISION OF CONSOLIDATED FINANCIAL STATEMENTS RELATED TO A SUBSEQUENT EVENT | 10 | SGX Company Announcements | ||
| 26.05. | TAKEDA PHARMACEUTICAL COMPANY LIMITED: GENERAL ANNOUNCEMENT::TAKEDA EXECUTIVE COMPENSATION OVERVIEW | 17 | SGX Company Announcements | ||
| 26.05. | TAKEDA PHARMACEUTICAL COMPANY LIMITED: GENERAL ANNOUNCEMENT::CEO-ELECT SHAREHOLDER LETTER 2026 | 7 | SGX Company Announcements | ||
| 26.05. | TAKEDA PHARMACEUTICAL COMPANY LIMITED: GENERAL ANNOUNCEMENT::OTHER ITEMS WHICH ARE PROVIDED ELECTRONICALLY FOR THE 150TH ORDINARY GENERAL MEETING OF SHAREHOLDERS | 3 | SGX Company Announcements | ||
| 26.05. | TAKEDA PHARMACEUTICAL COMPANY LIMITED: GENERAL ANNOUNCEMENT::NOTICE OF CONVOCATION OF THE 150TH ORDINARY GENERAL MEETING OF SHAREHOLDERS | 4 | SGX Company Announcements | ||
| 22.05. | Takeda Vows Appeal of $885M Jury Verdict in 'Pay-for-Delay' Antitrust Case | 5 | MedCity News | ||
| 22.05. | Should You Buy KROS After Q1 Miss and Takeda Deal Reset? | 9 | Zacks | ||
| 22.05. | Fierce Pharma Asia-Merck-Kelun ADC's triple wins; Tools in China licensing deals; Takeda's $885M antitrust loss | 8 | FiercePharma | ||
| 20.05. | Takeda faces $885m hit after landmark pay-for-delay lawsuit verdict | 19 | Pharmaceutical Technology | ||
| 19.05. | Takeda übertrifft Gewinnerwartungen im vierten Quartal 2025 | 28 | Investing.com Deutsch |
1 2 3 4 5 Weiter >>
162 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BASF | 48,340 | -0,38 % | BASF: EU macht Weg frei - doch diese Hürde bleibt | Die EU-Kommission hat grünes Licht für den Verkauf des BASF-Lackgeschäfts an den Finanzinvestor Carlyle gegeben - allerdings nur unter klaren Bedingungen. Wie die Behörde mitteilte, muss Carlyle zunächst... ► Artikel lesen | |
| K+S | 13,090 | -1,65 % | Aktien im Fokus - Bayer: Verkaufswelle - Aixtron, K+S, Rheinmetall, Siemens Energy und TeamViewer | Wo ist am Aktienmarkt etwas los, welche Themen interessieren Anleger derzeit besonders? Vor allem für Trader ist es wichtig zu wissen, wo "die Musik spielt" und welche Themen an der Börse aktuell besonders... ► Artikel lesen | |
| SGL CARBON | 4,795 | +0,31 % | SGL CARBON SE: Ruhe vor der nächsten Bewertung | ||
| LINDE | 448,40 | +0,81 % | Dividendenbekanntmachungen (04.06.2026) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) A-LIVING SMART CITY SERVICES CO LTD CNE100002RY5 0,0688 HKD 0,0075 EUR ADESSO SE DE000A0Z23Q5 - 0,78 EUR AGEAS SA/NV ADR US00844W2089 2... ► Artikel lesen | |
| HENKEL | 68,74 | +0,61 % | Henkel-Chef wirbt für Geduld mit Bundesregierung | Düsseldorf - Henkel-Chef Carsten Knobel hat weitere Zukäufe von Firmen angekündigt. Übernahmen seien ein wichtiger Bestandteil der Wachstumsstrategie, sagte der Vorstandsvorsitzende des Dax-Konzerns... ► Artikel lesen | |
| WACKER CHEMIE | 91,70 | -2,19 % | Aktien Frankfurt Ausblick: Wieder tiefer - Halbleiterrally weiter ausgebremst | FRANKFURT (dpa-AFX) - Bei schwachen Vorgaben aus Asien dürfte der Dax seine Vortagesgewinne am Freitag wieder abgeben. Knapp eine Stunde vor Handelsbeginn signalisierte der X-Dax als außerbörslicher... ► Artikel lesen | |
| LANXESS | 15,180 | -1,49 % | Lanxess-Aktie // Nur ein Rabatt-Kauf ergibt Sinn | Keinen schönen Anblick bietet der Chart der Lanxess-Aktie (547040). Denn seit Jahren geht es kontinuierlich abwärts. Der Kunststoff-Konzern leidet einerseits unter den hohen Energiekosten, andererseits... ► Artikel lesen | |
| COVESTRO | 60,00 | +0,17 % | EQS-RPT: Covestro AG: Veröffentlichung gemäß § 111c AktG mit dem Ziel der europaweiten Verbreitung | EQS Mitteilung über Geschäfte mit nahestehenden Personen: Covestro AG
/ Veröffentlichung gemäß § 111c AktG
Covestro AG: Veröffentlichung gemäß § 111c AktG mit dem Ziel der europaweiten... ► Artikel lesen | |
| FUCHS | 39,240 | +1,71 % | Aktien - Shortseller-Positionen: Fuchs, Heidelberger Druck, K+S, LPKF, TeamViewer, Vonovia | Wer Aktien leerverkauft, also auf fallende Kurse spekuliert (Shortselling), unterliegt bestimmten Transparenzpflichten. Aktuelle Meldungen über Leerverkäufe geben Aufschluss darüber. Diese Vorgaben... ► Artikel lesen | |
| SYMRISE | 81,42 | +0,25 % | EQS-PVR: Symrise AG: Veröffentlichung gemäß § 40 Abs. 1 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Stimmrechtsmitteilung: Symrise AG
Symrise AG: Veröffentlichung gemäß § 40 Abs. 1 WpHG mit dem Ziel der europaweiten Verbreitung
03.06.2026 / 11:04 CET/CEST
Veröffentlichung... ► Artikel lesen | |
| AIR LIQUIDE | 169,60 | -0,48 % | Air Liquide investiert 200 Mio Euro in Südkorea - liefert Gase für KI-Chipfertigung | DJ Air Liquide investiert 200 Mio Euro in Südkorea - liefert Gase für KI-Chipfertigung
Von Nina Kienle
DOW JONES--Air Liquide wird fast 200 Millionen Euro in Südkorea investieren. Das französische... ► Artikel lesen | |
| ALBEMARLE | 129,25 | -2,38 % | Sienna Resources baut Multi-Metall-Explorationsportfolio in aufstrebenden nordamerikanischen Bergbaudistrikten auf | ||
| HEXAGON COMPOSITES | 0,795 | -3,23 % | Hexagon Composites ASA: Approval of prospectus | NOT FOR RELEASE, PUBLICATION OR DISTRIBUTION, IN WHOLE OR IN PART DIRECTLY OR INDIRECTLY, IN OR INTO AUSTRALIA, CANADA, JAPAN, HONG KONG, SOUTH AFRICA OR THE UNITED STATES OR ANY OTHER JURISDICTION... ► Artikel lesen | |
| BRENNTAG | 55,10 | -0,25 % | GOLDMAN SACHS stuft Brenntag auf 'Buy' | NEW YORK (dpa-AFX Analyser) - Die US-Investmentbank Goldman Sachs hat das Kursziel für Brenntag von 71 auf 73 Euro angehoben und die Einstufung auf "Buy" belassen. Die Dauer der positiven Preis- und... ► Artikel lesen | |
| SQM | 65,00 | -0,91 % | Wolfram statt Lithium? Warum Almonty, die US-Verteidigung und Cosmos Exploration plötzlich Teil derselben Rohstoffdebatte sind |
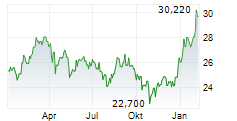
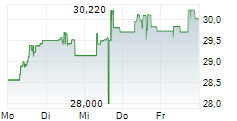
Das Unternehmen TAKEDA PHARMACEUTICAL CO LTD kann der Branche Chemie zugeordnet werden. Mit einer aktuellen Kursveränderung von -0,500 (-1,81 %) liegt der Kurs bei 27,190 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu TAKEDA PHARMACEUTICAL CO LTD finden Sie auf Wallstreet Online.