NEW BRUNSWICK (dpa-AFX) - The FDA handed out a string of approvals in late February and early March, granting regulatory clearances across several therapeutic areas - from oncology and rare genetic disorders to growth deficiencies.
Let's take a look at the treatments that recently secured the regulatory nod.
Boehringer Ingelheim Pharmaceuticals Inc.
On February 26, 2026, the FDA granted accelerated approval to Boehringer Ingelheim's Hernexeos as a first-line treatment for adult patients with advanced non-small cell lung cancer (NSCLC) whose tumors harbor HER2 tyrosine kinase domain-activating mutations, confirmed by an FDA-authorised test.
This approval represents a major milestone, making Hernexeos the first targeted therapy available for HER2-mutant advanced NSCLC in the first-line setting. HER2 (ERBB2) mutations occur in approximately 2-4% of NSCLC cases and are associated with a poor prognosis and higher incidence of brain metastases.
In August 2025, the FDA had granted accelerated approval for HERNEXEOS in patients with HER2-mutant advanced NSCLC who had received prior treatment.
Ascendis Pharma A/S (ASND)
Ascendis secured accelerated FDA approval for once-weekly Yuviwel for children with achondroplasia aged 2 years and older on February 27, 2026.
Achondroplasia is a rare genetic disorder affecting bone growth, often leading to skeletal dysplasia. People with this condition often face a higher risk of complications involving the muscles, nervous system, and heart or lungs.
Voxzogo, developed by BioMarin Pharmaceutical Inc. (BMRN), was the first FDA-approved therapy for children with achondroplasia. It received approval in November 2021 for pediatric patients five years and older and accelerated approval for children under five in October 2023. Voxzogo is administered as a once-daily subcutaneous injection.
Yuviwel is the first and only approved achondroplasia therapy to provide continuous systemic exposure to C-type natriuretic peptide (CNP) over the weekly dosing interval, offering a unique once-weekly treatment option compared with Voxzogo, which requires once-daily administration.
BioMarin Pharmaceutical Inc. (BMRN)
BioMarin secured FDA approval to expand the indication of Palynziq to include pediatric patients 12 years of age and older with phenylketonuria on Feb.27, 2026.
Phenylketonuria (PKU), also known as phenylalanine hydroxylase (PAH) deficiency, is a genetic disorder marked by elevated levels of the essential amino acid phenylalanine (Phe) in the bloodstream. Since the PAH enzyme is necessary for the normal breakdown of phenylalanine, its deficiency causes phenylalanine to accumulate in the blood. Excess Phe is toxic to the brain and can lead to serious complications, including profound intellectual disability, seizures, tremors, behavioral disturbances, and psychiatric symptoms.
Palynziq is the only enzyme substitution therapy approved to reduce blood phenylalanine (Phe) concentrations in people with PKU. The drug was approved for the treatment of adults with phenylketonuria who have uncontrolled blood Phe concentrations greater than 600 micromol/L in 2018.
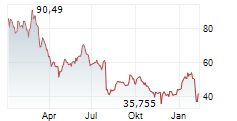
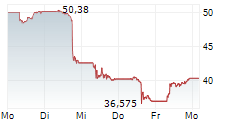
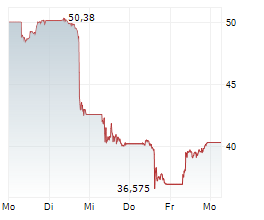
Since its approval in 2018, Palynziq has seen steady growth in sales. It generated $433 million in revenue in full-year 2025, compared to $355 million in 2024.
Novo Nordisk A/S (NVO)
On February 27, 2026, Novo Nordisk received FDA approval for three new indications for Sogroya, a long-acting growth hormone administered once weekly at 5 mg, 10 mg, or 15 mg. The expanded label now includes children aged 2.5 years and older with Idiopathic Short Stature (ISS), short stature born Small for Gestational Age (SGA) with no catch-up growth by 2 years of age, and growth failure associated with Noonan Syndrome (NS).
Previously, Sogroya was approved in August 2020 for the treatment of adults with growth hormone deficiency (GHD) and in April 2023 for children with GHD.
For more than four decades, treatment for growth disorders has relied on daily growth hormone injections, requiring children and caregivers to manage 365 doses each year. This demanding routine often poses challenges for adherence. A once-weekly alternative like Sogroya could ease the burden and improve compliance for families, according to the company.
Johnson & Johnson (JNJ)
On March 5, 2026, the FDA approved Tecvayli plus Darzalex Faspro for the treatment of adults with relapsed or refractory multiple myeloma (RRMM) who have received at least one prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent.
Tecvayli, as monotherapy, received accelerated approval from the FDA in October 2022 as an off-the-shelf (or ready-to-use) antibody that is administered as a subcutaneous treatment for adult patients with relapsed or refractory multiple myeloma (RRMM) who have received at least four prior lines of therapy, including a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 antibody.
Darzalex Faspro, also from the stable of JNJ, is already approved as monotherapy and in combinations for 11 indications in multiple myeloma, four of which are for frontline treatment in newly diagnosed patients who are transplant eligible or ineligible.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News