LONDON (dpa-AFX) - Daiichi Sankyo (4568.T) and AstraZeneca's (AZN, AZN.L, ZEG.DE, AZN.ST) supplemental Biologics License Application for ENHERTU has been accepted and granted Priority Review in the U.S. for the treatment of adult patients with HER2 positive breast cancer who have residual invasive disease after neoadjuvant HER2 targeted treatment. The sBLA is being reviewed under Project Orbis.
ENHERTU is a specifically engineered HER2 directed DXd antibody drug conjugate discovered by Daiichi Sankyo and being jointly developed and commercialized by Daiichi Sankyo and AstraZeneca. Regulatory submissions for ENHERTU based on DESTINY-Breast05 are under review in the EU and Japan.
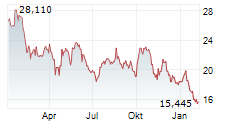
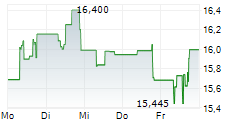
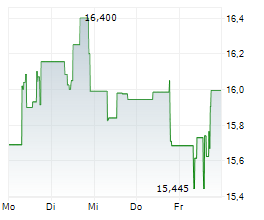
Daiichi Sankyo shares are currently trading at 2,849 yen, down 0.63%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News