LONDON (dpa-AFX) - AstraZeneca (AZN, AZN.L, ZEG.DE, AZN.ST) said Alexion, AstraZeneca Rare Disease's Koselugo has been approved in Canada for the treatment of adult patients with neurofibromatosis type 1 who have symptomatic, inoperable plexiform neurofibromas. The approval by Health Canada was based on positive results from KOMET.
Koselugo has been recently approved in the US, EU, Japan and other countries for the treatment of adult patients with neurofibromatosis type 1 who have symptomatic, inoperable plexiform neurofibromas based on data from the KOMET Phase III trial, and additional regulatory reviews are ongoing.
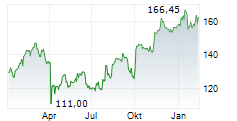
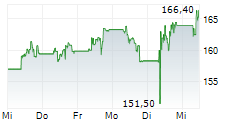
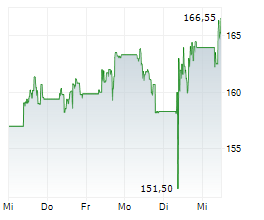
In pre-market trading on NYSE, AstraZeneca shares are down 2.78 percent to $188.92.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News