NEW YORK CITY (dpa-AFX) - Pfizer Inc. (PFE) on Thursday announced positive topline results from the Phase 3 TALAPRO-3 study of TALZENNA in combination with XTANDI in people with homologous recombination repair (HRR) gene-mutated metastatic castration-sensitive prostate cancer.
The study met its primary endpoint, with the combination demonstrating a statistically significant and clinically meaningful improvement in radiographic progression-free survival compared to placebo plus XTANDI. The results exceeded the pre-specified hazard ratio target of 0.63, with most patients remaining progression-free at the time of analysis.
The treatment also showed consistent benefits in patients with both BRCA and non-BRCA HRR gene alterations.
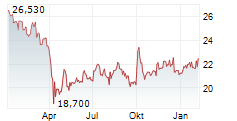
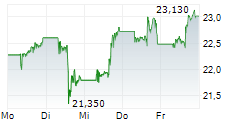
Pfizer shares closed at $27.32 on Wednesday, down 0.47%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News