LONDON (dpa-AFX) - GSK plc (GSK), Thursday announced that the US Food and Drug Administration has approved Lynavoy for the treatment of cholestatic pruritus in adult patients with primary biliary cholangitis, making it the first medicine approved in the US for this indication.
The approval is supported by data from the global GLISTEN phase III trial which met both primary and key secondary endpoints, showing significant, rapid and sustained improvements in cholestatic pruritus and itch-related sleep interference versus placebo.
Kaivan Khavandi, SVP, R&D Head Respiratory, Immunology & Inflammation, and Head of GSK Translational & Development Sciences, GSK, said, 'This is the first liver medicine from our pipeline to receive approval, underscoring our commitment to developing meaningful innovation across the spectrum of liver disease.'
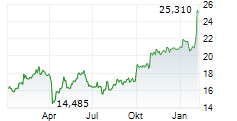
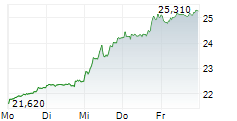
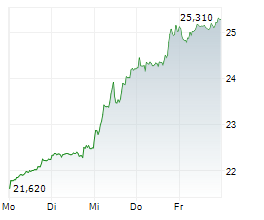
In the pre-market hours, GSK is trading at $51.70, down 0.71 percent on the NYSE.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News