NEW YORK CITY (dpa-AFX) - Pfizer (PFE) and Valneva SE (VALN) announced topline results from the Phase 3 VALOR Vaccine Against Lyme for Outdoor Recreationists trial of its investigational 6-valent OspA-based Lyme disease vaccine candidate PF-07307405 demonstrating: in the pre-specified analyses: efficacy of 73.2% from 28 days post-dose 4 in reducing the rate of confirmed Lyme disease cases compared to the placebo arm, efficacy of 74.8% from 1-day post-dose 4 in reducing the rate of confirmed Lyme disease cases compared to the placebo arm.
Fewer than anticipated Lyme disease cases were accrued over the study period, and the pre-determined statistical criterion was not met in the first pre-specified analysis. Given the clinically meaningful efficacy and the fact that the 95% confidence interval lower bound was above 20 in the second pre-specified analysis, Pfizer is planning submissions to regulatory authorities.
The companies entered into a collaboration and license agreement in April 2020 for the co-development of PF-07307405 and for Pfizer to manufacture and commercialize PF-07307405, assuming regulatory success.
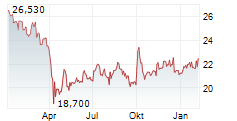
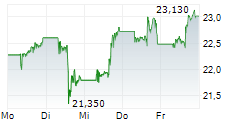
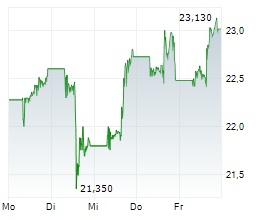
In pre-market trading on NasdaqGS, Valneva shares are down 35 percent to $6.70.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News