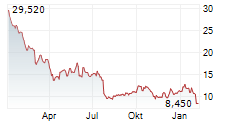
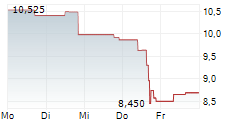
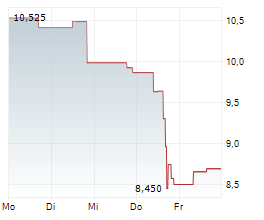
WASHINGTON (dpa-AFX) - Novocure (NVCR) announced Thursday positive results from the Phase 2 PANOVA-4 trial of Tumor Treating Fields (TTFields) therapy concomitant with atezolizumab (Tecentriq), gemcitabine and nab-paclitaxel (gem/nab-pac) as a first-line treatment for metastatic pancreatic ductal adenocarcinoma (mPDAC).
PANOVA-4 met its pre-specified primary endpoint, achieving a statistically significant improvement in disease control rate (DCR) compared to the DCR reported in the Phase 3 MPACT study used as the historical control.
The DCR in patients treated with TTFields therapy concomitantly with atezolizumab and gem/nab-pac was 74.4% compared to a DCR of 48% in patients receiving gem/nab-pac alone in the historical control.
In the PANOVA-4 trial, DCR was defined as the proportion of patients who had either stable disease (SD) for at least 16 weeks or confirmed partial response (PR) or complete response (CR) according to the Response Evaluation Criteria in Solid Tumours (RECIST v1.1).
Secondary endpoints in PANOVA-4 include objective response rate (ORR) and overall survival (OS).
The ORR in patients treated with TTFields therapy concomitantly with atezolizumab and gem/nab-pac was 34.6% and median OS was 9.7 months. Additional secondary endpoints were progression-free survival, one-year survival rate, progression-free survival at six months, duration of response, and rate of patients with treatment emergent adverse events.
The company said it looks forward to evaluating the full results from PANOVA-4 as it advances TTFields therapy as a treatment for metastatic pancreatic cancer.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News