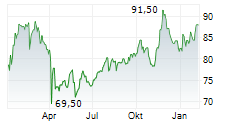
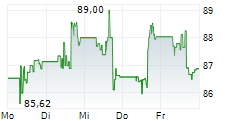
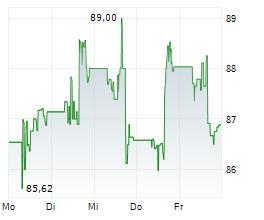
DUBLIN (dpa-AFX) - Healthcare technology company Medtronic plc (MDT) announced Friday U.S. Food and Drug Administration (FDA) clearance of the Stealth AXiS surgical system for cranial and ear, nose, and throat (ENT) procedures.
Cleared for spine procedures in February, the Stealth AXiS surgical system unifies surgical planning, navigation, and robotics into a single intelligent system to support surgeons with greater precision and real-time insights during complex procedures.
A defining capability of the platform is its AI-enabled architecture, which supports advanced planning and visualization before, during, and after surgery.
For cranial procedures, the system includes AI-based automatic tractography that helps surgeons generate patient-specific brain maps and visualize critical neural pathway associated with essential brain functions.
The platform also integrates intraoperative ultrasound through GE HealthCare's bkActiv, providing additional real-time imaging during procedures.
For ENT procedures, the Stealth AXiS surgical system delivers high-precision navigation and advanced visualization tailored to the intricate anatomy of the sinuses and skull base.
Additionally, the system's flexible architecture is designed to support continuous software innovation, expanded capabilities, and deeper ecosystem connectivity to help shape the next era of surgery.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News