NEW BRUNSWICK (dpa-AFX) - Johnson & Johnson (JNJ) announced new 52-week Phase 3 data from the ICONIC-ADVANCE 1 and 2 and ICONIC-LEAD studies evaluating ICOTYDE (icotrokinra) in moderate-to-severe plaque psoriasis. ICOTYDE is the first and only targeted oral peptide that blocks the IL-23 receptor.
In the ICONIC-ADVANCE studies, ICOTYDE achieved high rates of complete skin clearance (PASI 100) through Week 52. Rates increased from 41% to 49% in ADVANCE 1 and from 33% to 48% in ADVANCE 2 between Week 24 and Week 52. Patients who switched from placebo to ICOTYDE at Week 16 achieved similar clearance rates by Week 52 (50% and 43% in ADVANCE 1 and 2, respectively). The safety profile remained consistent with earlier data, with no new safety signals identified. Overall adverse event and infection rates were lower than those observed with deucravacitinib through Week 24.
In adolescents, ICOTYDE demonstrated sustained efficacy and safety in the ICONIC-LEAD study. Nearly 60% of patients achieved complete skin clearance at Week 52 (57% PASI 100, 61% IGA 0). Additionally, 86% achieved PASI 90 response at one year, with 92% maintaining that response from Week 24 to Week 52. No increase in adverse events was observed over the year-long treatment period.
These findings reinforce ICOTYDE's potential as a durable, safe, and effective oral therapy for both adults and adolescents with moderate-to-severe plaque psoriasis.
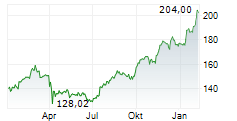
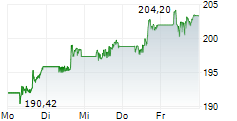
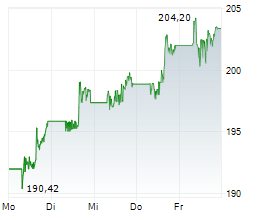
JNJ closed Friday's regular trading at $240.45 up $1.21 or 0.51%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News