LEVERKUSEN (dpa-AFX) - Bayer (BYR.L, BAYRY.PK, BAYZF.PK, BAY.MI, BAYN.DE) announced that the European Commission has granted approval in the European Union for Kerendia or finerenone, a selective, non-steroidal mineralocorticoid receptor antagonist for the treatment of adults with heart failure with left ventricular ejection fraction ?40%. In the EU, Kerendia is now indicated for the treatment of symptomatic chronic heart failure with LVEF ?40% in adults, expanding its use beyond the existing indication in patients with chronic kidney disease associated with type 2 diabetes.
The approval by the European Commission is based on the positive results from the pivotal Phase III FINEARTS-HF study.
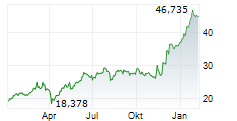
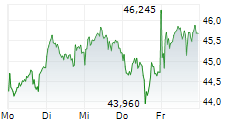
At last close, Bayer shares were trading at 38.26 euros.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News