PETAH TIKVA (dpa-AFX) - Teva Pharmaceutical Industries Limited (TEVA), Monday announced that the U.S. Food and Drug Administration has approved PONLIMSI as a biosimilar to Prolia, to treat a variety of debilitating bone conditions.
The FDA approval was based on a totality of evidence, including analytical and clinical data demonstrating similar efficacy, safety, and immunogenicity profile as the reference product, Prolia.
Also, Teva's applications for a proposed biosimilar candidate to Xolair have been accepted by both the U.S. FDA and the European Medicines Agency.
Yolanda Tibbe, Global Head of Biosimilars at Teva, commented, 'To receive U.S. FDA approval of PONLIMSI and the filing acceptances of our proposed biosimilar candidate to Xolair in the U.S. and Europe truly underscores the strength of our expanding global biosimilar portfolio and reaffirms our commitment to expand treatment options for patients.'
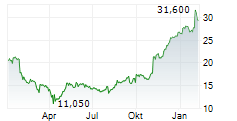
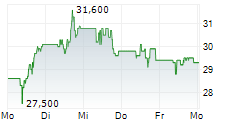
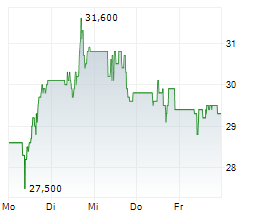
In the pre-market hours, TEVA is trading at $28.61, down 0.47 percent on the New York Stock Exchange.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News