BRUSSELS/FRANKFURT/PARIS (dpa-AFX) - GSK plc (GSK, GSK.L) announced that China's National Medical Products Administration has approved Exdensur or depemokimab as an add-on therapy with intranasal corticosteroids for the treatment of adult patients with CRSwNP for whom therapy with systemic corticosteroids and/or surgery do not provide adequate disease control. The approval is based on data from the ANCHOR-1 and ANCHOR-2 phase III trials.
Exdensur has also been approved in the US and China for the treatment of severe asthma, as well as in Japan, the EU, and UK for the treatment of severe asthma and CRSwNP.
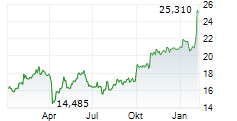
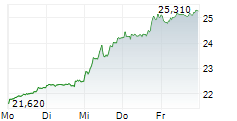
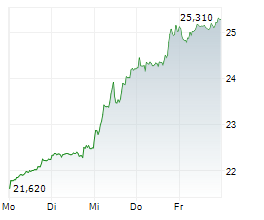
At last close, GSK plc shares were trading at 2,097.00 pence, down 2.19%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News