KENILWORTH (NJ) (dpa-AFX) - Daiichi Sankyo (4568.T) and Merck's (MRK) Biologics License Application for ifinatamab deruxtecan has been accepted and granted Priority Review by the FDA for the treatment of adult patients with extensive-stage small cell lung cancer with disease progression on or after platinum-based chemotherapy. The PDUFA date, the FDA action date for its regulatory decision, is October 10, 2026.
Ifinatamab deruxtecan is a specifically engineered, B7-H3 directed DXd antibody drug conjugate discovered by Daiichi Sankyo and being jointly developed by Daiichi Sankyo and Merck.
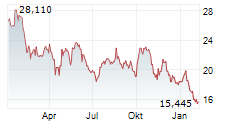
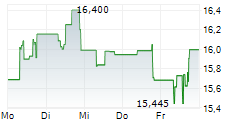
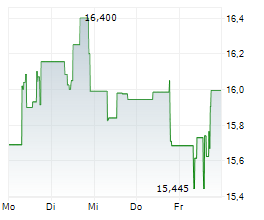
Daiichi Sankyo shares are trading at 2,732 yen, down 1.48%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News