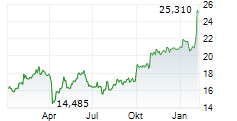
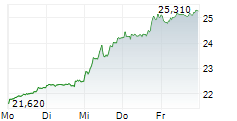
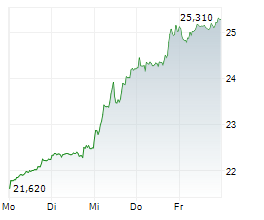
LONDON (dpa-AFX) - GSK plc (GSK) on Monday said China's National Medical Products Administration has approved Blenrep in combination with bortezomib and dexamethasone for the treatment of adults with relapsed or refractory Multiple Myeloma who have received at least one prior line of therapy.
The approval is supported by data from the pivotal Phase III DREAMM-7 trial, which showed statistically significant and clinically meaningful improvements in progression-free survival and overall survival for the Blenrep combination compared with a daratumumab-based regimen with bortezomib and dexamethasone.
GSK said the safety and tolerability profile of the Blenrep combination was broadly consistent with the known profiles of the individual medicines.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News