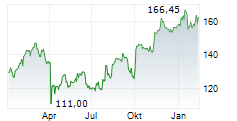
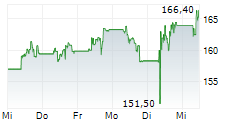
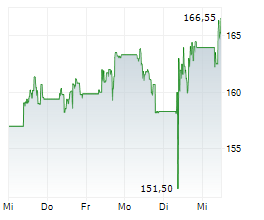
LONDON (dpa-AFX) - AstraZeneca PLC (AZN) on Tuesday said an interim analysis of its Phase III I CAN study showed that Ultomiris met the primary endpoint in adults with immunoglobulin A nephropathy.
The study demonstrated a statistically significant and clinically meaningful reduction in proteinuria at week 34.
The primary endpoints include change from baseline in proteinuria, measured by 24-hour urine protein creatinine ratio at week 34, and change in estimated glomerular filtration rate at week 106, to be assessed at the final analysis.
Ultomiris is already approved in the U.S., EU, Japan and other markets for the treatment of certain patients with paroxysmal nocturnal haemoglobinuria and atypical haemolytic uraemic syndrome.
It is also approved in multiple regions for certain adults with generalised myasthenia gravis and neuromyelitis optica spectrum disorder.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News