- Mangaciclanol, if approved, could transform contrast-enhanced MR imaging by offering an alternative to or even replacing gadolinium-based MRI agents
- A manganese-based MRI contrast agent could address concerns with gadolinium-based contrast agents around retention in the body, security of supply, and the environment, with mangaciclanol's clinical development under FDA Fast Track designation
- The program demonstrates GE HealthCare's commitment to advancing novel imaging agents to address unmet patient needs
GE HealthCare (Nasdaq: GEHC) today announced the first patient has been dosed in the international, multi-center Phase 2/3 LUMINA clinical trial of its manganese-based magnetic resonance imaging (MRI) contrast agent, mangaciclanol, at Mayo Clinic in Rochester, Minnesota. Mangaciclanol, if approved, could offer an alternative to or even replace gadolinium-based MRI contrast agents, the current standard of care. The investigational agent has been granted Fast Track designation by the U.S. Food and Drug Administration (FDA) for use in adults and pediatric patients aged 2 years and older with MRI to detect and visualize lesions with abnormal vascularity in the central nervous system and the body. FDA Fast Track designation expedites the review of new therapeutics and medical imaging agents that have the potential to address significant unmet patient needs.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260423262096/en/
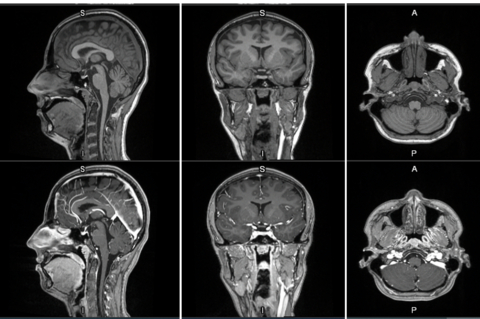
First-in-human contrast enhanced MRI images with GE HealthCare's mangaciclanol
MRI contrast agents enhance the visualization of abnormal structures or lesions during imaging procedures and help clinicians better distinguish between healthy and diseased tissue. Mangaciclanol is intended for general-purpose MR imaging and demonstrates comparable relaxivity (the ability to enhance signal intensity) to market-leading gadolinium-based agent, gadobutrol, with early clinical images suggesting similar diagnostic capability. Unlike gadolinium, which is a rare-earth metal, manganese is present in our food, and is an endogenous element, naturally occurring and autoregulated in the body. The macrocyclic 'cage-like' structure of mangaciclanol lessens the possibility of retention.
Approximately one-third of global MRI procedures require a contrast agent for effective diagnosis,1 with around 65 million gadolinium contrast enhanced procedures globally each year.2 As a rare-earth element, gadolinium supply is largely dependent on mining and processing infrastructure in China. Manganese, however, is abundantly available from multiple countries, including South Africa, Australia and Gabon, reducing the risk of supply challenges impacting patient care. Furthermore, as manganese is naturally found in water sources, mangaciclanol could also reduce environmental concerns associated with post-patient excreted contrast media in groundwater.
Dr Jit Saini, Chief Medical Officer, Pharmacutical Diagnostics, GE HealthCare, said, "Existing gadolinium-based contrast agents carry safety language associated with gadolinium retention. In comparison, mangaciclanol could offer an alternative for broad patient groups, including vulnerable patients and those requiring multiple scans, while still offering similar diagnostic performance. The FDA Fast Track designation recognizes the potential significance of mangaciclanol and aligns with our focus on advancing its development for patients."
"As demand for diagnostic imaging continues to rise, we continue to advance our imaging agent pipeline to better meet the needs of patients," said Peter Arduini, President and CEO, GE HealthCare. "This clinical milestone builds on GE HealthCare's leadership in contrast media, as mangaciclanol has the potential to transform the MR imaging market and strengthen the resiliency of its supply chain."
Phase 1 results for mangaciclanol showed the investigational agent was well tolerated in a first in human trial with no serious adverse events, no dose limiting toxicities, nor clinically relevant findings reported.
GE HealthCare's Pharmaceutical Diagnostics unit is a global leader in imaging agents used to support 140 million patient procedures per year globally, equivalent to four procedures every second. For more than 40 years, GE HealthCare contrast media has been routinely used across MRI, X-ray/CT and ultrasound to enhance clinical images and support diagnosis.
Mangaciclanol is in clinical development and currently not approved for use.
About GE HealthCare Technologies Inc.
GE HealthCare is a leading global healthcare solutions provider of advanced medical technology, pharmaceutical diagnostics, and AI, cloud and software solutions that help clinicians tackle the world's most complex diseases. Serving patients and providers for 130 years, GE HealthCare is delivering bold innovations designed for the next era of medicine across its Imaging, Advanced Visualization Solutions, Patient Care Solutions, and Pharmaceutical Diagnostics segments to help clinicians deliver more personalized, precise patient care. We are a $20.6 billion business with approximately 54,000 colleagues working to create a world where healthcare has no limits.
GE HealthCare is proud to be among 2026 Fortune World's Most Admired Companies.
Follow us on LinkedIn, Facebook, Instagram, or visit our website for our latest news and perspectives.
1 Contrast Agents of Magnetic Resonance Imaging and Future Perspective. Nanomaterials. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC10343825/ [europepmc.org]
2 Recent Developments and Future Perspectives in Magnetic Resonance Imaging and Computed Tomography Contrast Media. Frontiers in Radiology. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC12871431/
View source version on businesswire.com: https://www.businesswire.com/news/home/20260423262096/en/
Contacts:
GE HealthCare Media Contact:
David Morris
M: +44 7920 591370
david.j.morris@gehealthcare.com