NEW BRUNSWICK (dpa-AFX) - Johnson & Johnson (JNJ) announced the FDA has granted Priority Review to the supplemental Biologics License Application for IMAAVY as treatment for people living with warm autoimmune hemolytic anemia. The company noted that IMAAVY is the first therapy to receive FDA Priority Review for this condition. The FDA's decision is supported by results from the pivotal Phase 2/3 ENERGY study.
Leonard Dragone, Disease Area Leader, Autoantibody and Rheumatology, Johnson & Johnson, said: 'This designation highlights both the serious, life-threatening nature of wAIHA and the potential for IMAAVY, if approved, to help address a critical unmet need by delivering clinically meaningful outcomes for patients.'
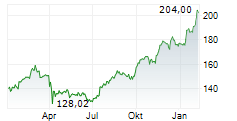
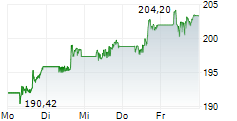
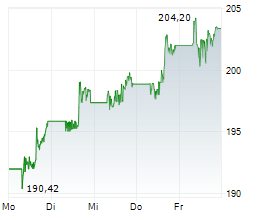
In pre-market trading on NYSE, Johnson & Johnson shares are down 0.03 percent to $227.38.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News