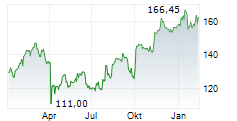
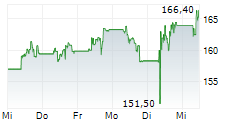
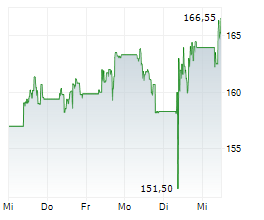
LONDON (dpa-AFX) - AstraZeneca PLC (AZN) on Tuesday said its triple-combination therapy Breztri Aerosphere has been approved by the U.S. Food and Drug Administration for the maintenance treatment of asthma in patients aged 12 years and older.
The therapy is a single inhaler combining an inhaled corticosteroid/long-acting beta2-agonist (ICS/LABA) with a long-acting muscarinic antagonist (LAMA).
The approval was based on Phase III KALOS and LOGOS trials, where Breztri (budesonide/glycopyrrolate/formoterol fumarate) showed statistically significant and clinically meaningful improvements in lung function compared with dual-combination ICS/LABA treatments across a broad asthma population.
Breztri was already approved in the U.S. in 2020 for the treatment of adults with chronic obstructive pulmonary disease (COPD).
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News