NEW YORK CITY (dpa-AFX) - Pfizer Inc. (PFE) on Wednesday reported positive topline results from the Phase 3 MagnetisMM-5 study evaluating ELREXFIO in adults with relapsed or refractory multiple myeloma who had received at least one prior line of treatment.
The study met its primary endpoint, showing a statistically significant improvement in progression-free survival compared with standard-of-care daratumumab plus pomalidomide and dexamethasone.
The safety and tolerability profile of ELREXFIO was consistent with prior studies, with most treated patients remaining progression-free at the interim analysis. The trial is ongoing to assess overall survival, which was not yet mature at the time of analysis.
ELREXFIO is already approved in more than 35 countries, including the United States and the European Union for certain patients with relapsed or refractory multiple myeloma.
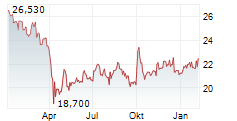
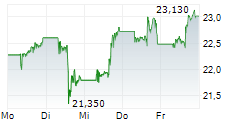
On Tuesday, Pfizer shares closed at $26.48, down 1.16%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News