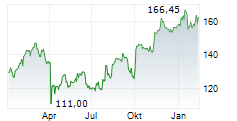
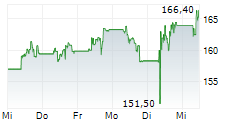
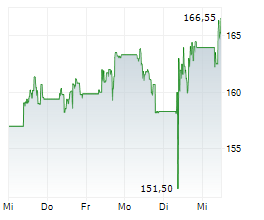
LONDON (dpa-AFX) - AstraZeneca (AZN, AZN.L, ZEG.DE, AZN.ST) said the FDA's Oncologic Drugs Advisory Committee or ODAC has recognised a favourable benefit risk profile for AstraZeneca's Truqap in combination with abiraterone and androgen deprivation therapy for the treatment of patients with PTEN-deficient metastatic hormone-sensitive prostate cancer, based on the CAPItello-281 Phase III trial. The Committee voted 7 to 1, with 1 abstaining.
In August 2025, the FDA accepted the sNDA for Truqap in combination with abiraterone and ADT. The ODAC provides the FDA with independent, expert advice on marketed and investigational medicines for use in the treatment of cancer. The FDA will consider the feedback as it reviews the submission.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News