Lund, Sweden - Clinical Laserthermia Systems AB (publ) ("CLS" or the "Company") today publishes its interim report for January - March 2026.
Highlights of First Quarter Interim Report 2026
During the first quarter, CLS continued to produce and deliver products, development, and support services to its partners, supporting the expansion of the installed base and adoption of its product portfolio.
- Through its partner in neurosurgery, CLS saw continued ClearPoint Prism® System installations and procedure growth in the U.S., reflecting underlying clinical and commercial momentum.
- However, CLS sales of Prism products towards its partner in neurosurgery were temporarily impacted by ongoing alignment discussions regarding the acceleration and scaling of the Prism business. As a result, revenue for the quarter amounted to SEK 1.3 million, a decrease of 72% compared to the same period last year.
- These discussions were successfully finalized after the end of the quarter in mid-April, establishing a clear plan and commitments for accelerating growth of the Prism business going forward.
- Following on the established growth plan and commitments, CLS received purchase orders totaling approximately SEK 23 million (€2.1 million) for its neurosurgery Prism business from its partner.
- CLS maintained strict cost control and improved operational efficiency. As a result, the decline in operating profit for the quarter, amounting to SEK 1.7 million compared to the same period in 2025, reflects the lower revenue and not an increase in operating expenses
- CLS continued its interaction with its notified body regarding the CE marking application for the ClearPoint Prism® Neuro Laser Therapy System for use with 1.5T and 3.0T MRI guidance. Regulatory clearance in Europe is expected during the second half of 2026 and is anticipated to support further growth acceleration.
Outlook 2026
With the agreement between CLS and its strategic partner in the US in place to accelerate the growth of the Prism business, CLS expects full-year 2026 revenue to show strong double-digit growth compared to 2025, with performance weighted toward the second half of the year.
- The Company has secured a meaningful portion of its expected short-term revenue in the committed and received purchase orders, providing a solid foundation for the year.
- Additional revenue is expected to be generated from an increased number of LITT procedures performed on the growing base of installed Prism systems, and continued commercial activity as the accelerated growth plan is implemented.
Revenue development remains dependent on the timing of system placements and the ramp-up of LITT procedure volumes and will therefore vary between quarters.
This outlook reflects the Company's current expectations based on available information and may be subject to change.
Summary of the Year-end report (relates to the Group)
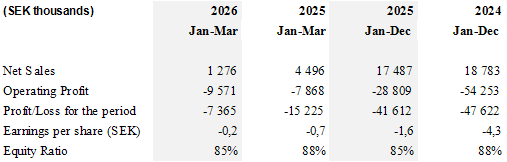
Comments from CEO
In the first quarter, sales were impacted by ongoing discussions with our neurosurgery partner regarding the conditions for scaling of the Prism business, reflecting a transition toward a significantly expanded commercial opportunity driven by the expanded FDA clearance for use with both 3.0T and 1.5T MRI guidance, our partner's increased capabilities and a growing LITT market.
While the discussions have impacted revenue in the short term, it is important to emphasize that the underlying fundamentals of our business remain strong. We continue to see solid clinical momentum and increasing adoption of neuro LITT procedures within neurosurgery, supported by updated clinical guidelines and established reimbursement.
Operationally, CLS remains well positioned. We continue to benefit from a structurally improved cost base, supporting a positive operational cash flow trend.
Following the end of the quarter, we announced two important milestones - the agreement with our strategic partner to accelerate the growth of the Prism business followed by purchase orders securing short-term revenue. This agreement establishes a clear and jointly committed plan within a long-term partnership framework extending through 2030.
This marks a clear shift for CLS - from a period of alignment and preparation into a phase focused on accelerated growth and scaling with more stability.
At the same time, we continue to execute our broader partnership strategy. The installation of the TRANBERG® system at Andros Clinics and the successful treatment of initial patients highlight continued clinical relevance and market interest in focal therapy of localized prostate cancer within urology.
Looking ahead
Looking ahead, we expect 2026 to be a transitional year, where the operational foundation for accelerated growth is established, with increasing commercial momentum as the year progresses.
Key priorities for the remainder of the year include:
- Supporting broader U.S. adoption of Prism under the expanded clearance
- Executing the accelerated growth plan together with our partner
- Advancing regulatory initiatives in Europe
- Continuing to develop and expand our strategic partnership model
- Maintaining a disciplined cost structure while enabling growth
As a result, we expect commercial activity and revenue to pick up in the coming quarters, with some variability between quarters depending on the timing of system placements and procedure ramp-up.
As mentioned in our earlier communication, significant unmet medical needs exist across several established and emerging indications addressed by the Prism product portfolio, including treatment of drug-resistant epilepsy, primary brain tumors, metastatic brain tumors, radiation necrosis, as well as future brain and spine applications.
To fully capture identified opportunities, CLS intends to invest in:
- Product scalability
- Regulatory expansion
- Working capital for accelerated growth
- Next-generation product development
- Expansion of its strategic partnership model
While the growth plan requires increased near-term investments, we believe it creates a stronger and more scalable path for CLS toward sustainable value creation. With a leading position in Neuro LITT, we will also be well positioned to expand into additional market segments.
With a clear focus on acceleration, scaling and long-term value creation we have updated our guidance on achieving positive cash flow from Q4 2026 to Q1 2028 and are evaluating financing alternatives intended at strengthening the Company's ability to execute on the growth plan.
With strong clinical momentum, lowered barriers to adoption, focused organization, and a strengthened partnership in neurosurgery, CLS is operationally well positioned to accelerate growth and expand access to minimally invasive laser therapy to a continuously growing patient population.
I would like to thank our employees, partners, and shareholders for your continued trust as we build CLS toward sustainable profitability and long-term value creation.
Dan J. Mogren, CEO
Clinical Laserthermia Systems AB
Significant events in the first quarter of 2026
- CLS announced the installation of the TRANBERG Thermal Therapy System at Andros Clinics in Amsterdam, through its distribution partner MTEC Company. The system is intended for ultrasound-guided focal laser ablation (FLA), a minimally invasive treatment option to radical prostatectomy for patients with localized prostate cancer.
Significant events after the end of the period
- CLS announced it has reached an agreement with its strategic partner on an alignment to accelerate the growth of the ClearPoint Prism® business in the U.S. The plan includes market expansion, product development, and revenue growth with strengthened long-term outlook. The commercial partnership runs through 2030, with an option for further extension, supporting CLS aim to become market leader in minimally invasive Laser Interstitial Thermal Therapy (LITT) within neurosurgery.
- CLS announced that it has received purchase orders totaling approximately SEK 23 million (€2.1 million) for its neurosurgery ClearPoint Prism® business from its strategic partner in neurosurgery. The orders represent the largest purchase order received to date for the Prism business and form part of the previously communicated agreement between the parties to accelerate the growth of the Prism business beginning in 2026.
For more information, please contact:
Dan J. Mogren, CEO Clinical Laserthermia Systems AB (publ)
Phone: +46 (0)705 90 11 40
E-mail: dan.mogren@clinicallaser.com
About CLS
Clinical Laserthermia Systems AB (publ), develops and sells TRANBERG® Thermal Therapy System and ClearPoint Prism® Neuro Laser Therapy System with sterile disposables, for minimally invasive treatment of cancer tumors and drug-resistant epilepsy. The products are marketed and sold through partners for image-guided laser ablation. CLS is headquartered in Lund, Sweden, with subsidiaries in Germany, the United States and a marketing company in Singapore. CLS is listed on Nasdaq First North Growth Market under the symbol CLS B. Certified adviser (CA) is FNCA Sweden AB.
For more information about CLS, please visit the Company's website: www.clinicallaser.se
This disclosure contains information that CLS is obliged to make public pursuant to the EU Market Abuse Regulation (EU nr 596/2014). The information was submitted for publication, through the agency of the contact person, on 12-05-2026 08:30 CET.