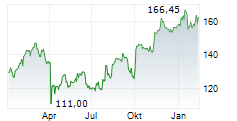
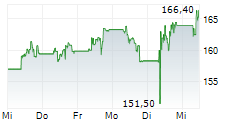
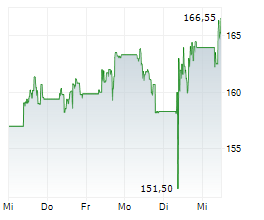
LONDON (dpa-AFX) - AstraZeneca plc (AZN) on Thursday said an interim analysis of the Phase III VOLGA trial showed that perioperative treatment with Imfinzi combined with enfortumab vedotin (EV) significantly improved event-free survival and overall survival in patients with muscle-invasive bladder cancer compared with standard of care.
The trial included patients who were ineligible for or declined cisplatin-based chemotherapy.
A combination of Imfinzi, Imjudo and EV also improved event-free survival, but overall survival results were not statistically significant at the interim analysis. The company said no new safety signals were identified.
Imfinzi is approved in more than 40 countries for patients with cisplatin-eligible muscle-invasive bladder cancer. Imfinzi combined with Bacillus Calmette-Guérin therapy for high-risk non-muscle-invasive bladder cancer is under regulatory review in the U.S., European Union, Japan and other countries. The drug is also being studied in locally advanced or metastatic disease in the Phase III NILE trial.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News