- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 13:30 | OS Therapies Says New Bone Cancer Data Strengthens Case For Regulatory Filings | 1 | Benzinga.com | ||
| 12:02 | OS Therapies: OS Therapies Achieves Statistically Significant 2.5-Year Overall Survival in Phase 2b Trial of OST-HER2 in Fully Resected Pulmonary Metastatic Osteosarcoma | 257 | Newsfile | 75% 2.5-year overall survival for OST-HER2 vs. 47% pooled historical control (p = 0.003), with no new patient deaths reported since the 2-year overall survival data (75% vs. 60%, p = 0.034)Updated... ► Artikel lesen | |
| Fr | OS Therapies: OS Therapies Announces the Publication of Four Articles on OST-HER2 and Osteosarcoma in Drug Discovery World | 418 | Newsfile | The Company confirms that it will release 2.5 year overall survival data from Phase 2b clinical trial OST-HER2 in the prevention or delay of recurrence in fully-resected pulmonary metastatic osteosarcoma... ► Artikel lesen | |
| 26.05. | OS Therapies: OS Therapies to Attend Upcoming Medical, Financial and Industry Conferences | 435 | Newsfile | ASCO 2026: 2.5-Year Overall Survival DataJefferies Healthcare Conference: Partnering & Investor meetingsBIO International Convention: Panel ParticipationMIB Agents Factor Osteosarcoma Conference:... ► Artikel lesen | |
| OS THERAPIES Aktie jetzt für 0€ handeln | |||||
| 18.05. | OS Therapies GAAP EPS of -$0.27 | 1 | Seeking Alpha | ||
| 18.05. | OS Therapies: OS Therapies Reports First Quarter 2026 Financials and Provides Business Update | 455 | Newsfile | $5.5 million financing that closed on April 2, 2026 together with $5.7 million in capital raised in Q1-2026, expected to provide Company with cash runway into 2027$4.5 million of accrued VAT refunds... ► Artikel lesen | |
| 18.05. | OS Therapies Inc - 10-Q, Quarterly Report | 1 | SEC Filings | ||
| 15.05. | OS Therapies: OS Therapies to Announce First Quarter 2026 Financials on Monday, May 18, 2026 | 411 | Newsfile | New York, New York and Rockville, Maryland--(Newsfile Corp. - May 15, 2026) - OS Therapies, Inc. (NYSE American: OSTX) ("OS Therapies" or "the Company"), the world leader in gene-edited, listeria-based... ► Artikel lesen | |
| 14.05. | OS Therapies: OS Therapies Appoints Industry Leader Dr. Craig Eagle as Chief Medical Advisor | 464 | Newsfile | Appointment strengthens medical, regulatory, and commercial leadership as Company prepares to complete early market access regulatory filings in the U.S., U.K, Europe and Australia in anticipation... ► Artikel lesen | |
| 12.05. | Reportable, Inc.: Stonegate Capital Partners Updates Coverage on OS Therapies Inc. (OSTX) | 382 | Newsfile | Dallas, Texas--(Newsfile Corp. - May 12, 2026) - OS Therapies Inc. (NYSE American: OSTX): Stonegate Capital Partners updates their coverage on OS Therapies Inc. (NYSE American: OSTX). OS Therapies... ► Artikel lesen | |
| 30.04. | OS Therapies Inc - 8-K, Current Report | - | SEC Filings | ||
| 30.04. | EMA begins rolling review of OS Therapies' osteosarcoma treatment | 1 | Investing.com | ||
| 30.04. | EMA startet fortlaufende Prüfung der Osteosarkom-Therapie von OS Therapies | 1 | Investing.com Deutsch | ||
| 30.04. | OS Therapies: OS Therapies Announces EMA Initiates Rolling Review of Conditional Marketing Authorization Application for OST-HER2 in the Prevention or Delay of Recurrence in Fully Resected Pulmonary Metastatic Osteosarcoma | 364 | Newsfile | Conference call scheduled for Thursday, April 30, 2026, at 8:30 am ET to review new OST-HER2 immune pharmacodynamic biomarker response (seroconversion) data and review regulatory successes validating... ► Artikel lesen | |
| 27.04. | OS Therapies: OS Therapies Schedules OST-HER2 Pharmacodynamic Response Biomarker Conference Call on April 30, 2026 at 8:30am ET | 356 | Newsfile | Regulatory feedback recent from April 2026 EU EMA and Australian TGA meetingsRemaining Q2-2026 regulatory meetings include two U.S. FDA meetings and one UK MHRA meeting, in addition to follow-up... ► Artikel lesen | |
| 16.04. | OS Therapies: OS Therapies Files New Patent Application Covering Biomarkers of the Immune Response to Listeria Monocytogenes | 400 | Newsfile | New patent application covers treatment-emergent immune signature related to 'turning cold tumors hot' and the activation of targeted cytotoxic cellular immune responses Company to host conference... ► Artikel lesen | |
| 13.04. | OS Therapies: OS Therapies Appoints Biotech Industry Luminary Robert "Bob" S. Langer, PhD as Strategic Advisor | 789 | Newsfile | Co-founder of more than 40 biotechnology companies, including 16 IPOs and 19 successful acquisitions Scientific and medical titan focused on driving innovation for human healthWill assist with listeria... ► Artikel lesen | |
| 08.04. | OS Therapies: OS Therapies Appoints Craig Eagle, MD as Strategic Advisor | 1.076 | Newsfile | Senior leadership roles at Guardant Health, Genentech and PfizerRegulatory, clinical and commercial expertise in therapeutics and biomarkersWill assist with osteosarcoma regulatory advice and oncology... ► Artikel lesen | |
| 02.04. | OS Therapies: OS Therapies Completes $5.25M Registered Direct Offering Primarily with Pre-Existing High-Net-Worth Investors | 819 | Newsfile | Company expects approximately $2 million in non-dilutive VAT refunds from wholly owned U.K. subsidiary in 2Q-26Company expects to receive approximately $2 million in non-dilutive R&D tax credits... ► Artikel lesen | |
| 31.03. | OS Therapies Inc - 8-K, Current Report | - | SEC Filings |
1 2 3 4 Weiter >>
61 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| CRISPR THERAPEUTICS | 47,085 | +1,07 % | CRISPR Therapeutics' Secret Weapon That Many Investors Are Overlooking | ||
| IMMUNIC | 13,055 | +0,35 % | EQS-News: Immunic AG: Immunic to Present Additional Phase 2 CALLIPER Trial Data for Vidofludimus Calcium at the CMSC Annual Meeting 2026, Reinforcing Its Potential in Progressive Multiple Sclerosis | Issuer: Immunic AG
/ Key word(s): Conference/Study results
Immunic to Present Additional Phase 2 CALLIPER Trial Data for Vidofludimus Calcium at the CMSC Annual Meeting 2026,... ► Artikel lesen | |
| PHIO PHARMACEUTICALS | 1,130 | 0,00 % | Phio Pharmaceuticals Corp.: Phio Pharmaceuticals Reports First Quarter 2026 Financial Results and Business Update | Key Development Agreements Secured for Nonclinical Toxicology and U.S. cGMP Clinical Supply ManufacturingKing of Prussia, Pennsylvania--(Newsfile Corp. - May 7, 2026) - Phio Pharmaceuticals Corp.... ► Artikel lesen | |
| ARCTURUS THERAPEUTICS | 7,785 | +0,19 % | Arcturus outlines 12-week ARCT-032 Phase II enrollment and extends cash runway beyond Q2 2028 | ||
| ANAVEX LIFE SCIENCES | 2,542 | -1,28 % | ANAVEX LIFE SCIENCES CORP - Struktur vor Impuls | ||
| ZEVRA THERAPEUTICS | 10,150 | +2,73 % | Zevra Therapeutics Announces Inducement Grants under Nasdaq Listing Rule 5635(c)(4) | ||
| PROTALIX BIOTHERAPEUTICS | 1,734 | -1,14 % | Protalix BioTherapeutics, Inc.: Protalix BioTherapeutics Reports First Quarter 2026 Financial and Business Results | Company to host conference call and webcast today at 8:00 a.m. EDT
Elfabrio commercial execution continues following European Commission approval of the 2 mg/kg... ► Artikel lesen | |
| GEOVAX LABS | 2,260 | +2,26 % | EQS-News: GeoVax, Inc.: GeoVax CEO Welcomes FIFA World Cup 2026 While Addressing Biothreat Readiness | EQS-News: GeoVax, Inc.
/ Key word(s): Science
GeoVax CEO Welcomes FIFA World Cup 2026 While Addressing Biothreat Readiness 01.06.2026 / 15:20 CET/CEST
The issuer is solely... ► Artikel lesen | |
| SCILEX | 7,750 | +11,51 % | Scilex Holding Company ("Scilex") Announces Its Board of Directors Approved a Dividend of Semnur Pharmaceuticals, Inc. Common Stock to Holders of Scilex Common Stock and other Eligible Equity Securities with a Record Date of June 1, 2026 | PALO ALTO, Calif., May 22, 2026 (GLOBE NEWSWIRE) -- Scilex Holding Company ("Scilex" or the "Company") (Nasdaq: SCLX), an innovative revenue-generating company focused on acquiring, developing, and... ► Artikel lesen | |
| INVIVYD | 1,210 | -0,82 % | Invivyd Announces Completion of Enrollment in Upsized DECLARATION Clinical Trial, a Phase 3 Pivotal Study of VYD2311, an Investigational Antibody to Prevent COVID | DECLARATION is a Phase 3, randomized, placebo-controlled clinical trial to evaluate the safety and efficacy of VYD2311 in the prevention of symptomatic COVID-19 versus placebo, at three months, from... ► Artikel lesen | |
| MEIRAGTX | 8,950 | +5,92 % | MeiraGTx Reports First Quarter 2026 Financial and Operational Results | Received FDA Breakthrough Therapy Designation for AAV2-hAQP1Reported positive three-year data from the Phase 1 AQUAx study of AAV2-hAQP1 for the treatment of grade 2/3 late radiation-induced xerostomiaEntered... ► Artikel lesen | |
| TENAYA THERAPEUTICS | 0,870 | 0,00 % | Tenaya Therapeutics, Inc. - 8-K, Current Report | ||
| UNICYCIVE THERAPEUTICS | 7,590 | -0,13 % | H.C. Wainwright reiterates Unicycive stock rating on manufacturing progress | ||
| SUMMIT THERAPEUTICS | 15,710 | 0,00 % | Cantor Fitzgerald Maintains Overweight Rating on Summit Therapeutics (SMMT) Amid Mixed Clinical Developments | ||
| ERASCA | 13,820 | 0,00 % | Erasca, Inc.: Erasca Reports First Quarter 2026 Business Updates and Financial Results | Robust monotherapy efficacy and generally well-tolerated safety results observed during dose escalation for ERAS-0015 in both KRAS G12X NSCLC and PDAC reinforce best-in-class potential across RAS-targeted... ► Artikel lesen |
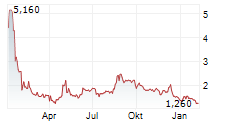
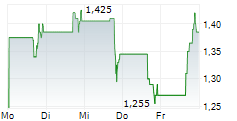
Das Unternehmen OS THERAPIES INC kann der Branche Biotechnologie zugeordnet werden. Mit einer aktuellen Kursveränderung von +0,005 (+0,26 %) liegt der Kurs bei 1,925 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu OS THERAPIES INC finden Sie auf Wallstreet Online.