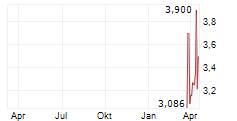- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 27.05. | XUANZHUBIO-B (02575): FORM OF PROXY FOR THE ANNUAL GENERAL MEETING | 3 | HKEx | ||
| 27.05. | XUANZHUBIO-B (02575): NOTICE OF THE 2025 ANNUAL GENERAL MEETING | 1 | HKEx | ||
| 27.05. | XUANZHUBIO-B (02575): CIRCULAR FOR THE 2025 ANNUAL GENERAL MEETING | 1 | HKEx | ||
| 26.05. | XUANZHUBIO-B (02575): PROPOSED AMENDMENT TO THE ARTICLES OF ASSOCIATION, ABOLITION OF THE SUPERVISORY COMMITTEE AND CHANGE OF BOARD COMPOSITION | - | HKEx | ||
| 12.05. | XUANZHUBIO-B (02575): NEXT DAY DISCLOSURE RETURN | 1 | HKEx | ||
| 12.05. | XUANZHUBIO-B Completes Full Circulation of H Shares | 1 | AASTOCKS | ||
| XUANZHU BIOPHARMACEUTICAL Aktie jetzt für 0€ handeln | |||||
| 12.05. | XUANZHUBIO-B (02575): INSIDE INFORMATION COMPLETION OF THE H SHARE FULL CIRCULATION BY THE COMPANY | 1 | HKEx | ||
| 27.04. | XUANZHUBIO-B (02575): LETTER TO NON-REGISTERED SHAREHOLDERS - REMINDER LETTER REGARDING THE ARRANGEMENT OF ELECTRONIC DISSEMINATION OF CORPORATE COMMUNICATIONS ... | 2 | HKEx | ||
| 27.04. | XUANZHUBIO-B (02575): LETTER TO REGISTERED SHAREHOLDERS - REMINDER LETTER REGARDING THE ARRANGEMENT OF ELECTRONIC DISSEMINATION OF CORPORATE COMMUNICATIONS ... | 1 | HKEx | ||
| 27.04. | XUANZHUBIO-B (02575): 2025 ENVIRONMENTAL, SOCIAL AND GOVERNANCE REPORT | - | HKEx | ||
| 27.04. | XUANZHUBIO-B (02575): 2025 ANNUAL REPORT | 1 | HKEx | ||
| 20.04. | XUANZHUBIO-B (02575): VOLUNTARY ANNOUNCEMENT RESULTS OF PRELIMINARY DATA FROM THE PHASE 1B FORTRESS CLINICAL STUDY OF NG-350A PRESENTED AT THE 2026 AACR | - | HKEx | ||
| 17.04. | XUANZHUBIO-B (02575): INSIDE INFORMATION APPROVAL BY THE STOCK EXCHANGE REGARDING THE APPLICATION FOR THE PROPOSED IMPLEMENTATION OF H SHARE FULL CIRCULATION | 1 | HKEx | ||
| 15.04. | XUANZHUBIO-B (02575): VOLUNTARY ANNOUNCEMENT APPROVAL TO INITIATE A PHASE III CLINICAL TRIAL FOR NEW INDICATION OF ANAPRAZOLE SODIUM | 1 | HKEx | ||
| 09.04. | XUANZHUBIO-B (02575): VOLUNTARY ANNOUNCEMENT - LICENSING AND SUPPLY AGREEMENT WITH BOSTON ONCOLOGY FOR BIREOCICLIB AND DIROZALKIB | 1 | HKEx | ||
| 30.03. | XUANZHUBIO-B (02575): ANNUAL RESULTS ANNOUNCEMENT FOR THE YEAR ENDED DECEMBER 31, 2025 | - | HKEx | ||
| 27.03. | XFRA NEW INSTRUMENTS AVAILABLE ON 27.03.2026 | 573 | Xetra Newsboard | The following instruments on XETRA do have their first trading 27.03.2026 Die folgenden Instrumente in XETRA haben ihren ersten Handelstag 27.03.2026
Aktien
1 SE0006887451 Corline Biomedical AB
2... ► Artikel lesen | |
| 24.03. | XUANZHUBIO-B (02575): VOLUNTARY ANNOUNCEMENT FINAL ANALYSIS RESULTS OF THE PHASE 3 CLINICAL STUDY (BRIGHT-2) OF BIREOCICLIB IN COMBINATION WITH FULVESTRANT ... | 3 | HKEx | ||
| 16.03. | XUANZHUBIO-B (02575): DATE OF BOARD MEETING | 1 | HKEx | ||
| 03.03. | XUANZHUBIO-B (02575): VOLUNTARY ANNOUNCEMENT MARKETING APPROVAL FOR THE THIRD INDICATION OF XUANYUENING (BIREOCICLIB TABLETS) FOR THE FIRST-LINE TREATMENT ... | - | HKEx |
1 2 Weiter >>
25 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| PRAXIS PRECISION MEDICINES | 258,08 | 0,00 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Vormatrigine Program Update | POWER1 Study in highly refractory patients with focal onset seizures did not meet its primary success measure Secondary measure, the 50% response rate, was met and seizure reduction during the second... ► Artikel lesen | |
| SUMMIT THERAPEUTICS | 14,860 | 0,00 % | Cantor Fitzgerald Maintains Overweight Rating on Summit Therapeutics (SMMT) Amid Mixed Clinical Developments | ||
| ERASCA | 13,480 | 0,00 % | Erasca, Inc.: Erasca Reports First Quarter 2026 Business Updates and Financial Results | Robust monotherapy efficacy and generally well-tolerated safety results observed during dose escalation for ERAS-0015 in both KRAS G12X NSCLC and PDAC reinforce best-in-class potential across RAS-targeted... ► Artikel lesen | |
| APOGEE THERAPEUTICS | 78,71 | 0,00 % | Weekly Buzz: AstraZeneca, JNJ Get FDA Nod; Eli Lilly To Acquire Curevo; APGE Advances Zumilokibart | NEW BRUNSWICK (dpa-AFX) - This week in the biotech space, there were regulatory approvals across the U.S. and Europe, collaborations, acquisitions, and positive data readouts spanning multiple... ► Artikel lesen | |
| RELAY THERAPEUTICS | 15,690 | -0,06 % | Relay Therapeutics Shares Jump 18% | ||
| COGENT BIOSCIENCES | 32,760 | 0,00 % | Cogent Biosciences, Inc.: Cogent Biosciences Announces Detailed Clinical Data from PEAK Phase 3 Trial with Bezuclastinib in Combination with Sunitinib in Gastrointestinal Stromal Tumors (GIST) at 2026 American Society of Clinical Oncology (ASCO) ... | Bezuclastinib combination is first treatment ever to demonstrate statistically significant advantage against active comparator in GIST patients; median PFS of 16.5 months versus 9.2 months (HR=0.50... ► Artikel lesen | |
| QIAGEN | 30,705 | -0,36 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| EDGEWISE THERAPEUTICS | 37,300 | +0,13 % | Servier to Acquire Edgewise Therapeutics' Muscular Dystrophy Business | Acquisition supports Servier's strategic ambition in rare neurology for patients with high unmet medical needs.Transaction includes sevasemten, a late-stage investigational treatment for... ► Artikel lesen | |
| TANGO THERAPEUTICS | 21,505 | +0,02 % | Tango Therapeutics, Inc.: Tango Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Highlights | First clinical data from PRMT5/RAS(ON) combination trial to be presented in 2026 Cash position of $380 million as of March 31, 2026, with runway into 2028 beyond anticipated key data inflection points... ► Artikel lesen | |
| OLEMA PHARMACEUTICALS | 10,690 | 0,00 % | Olema Oncology Announces Clinical Trial Collaboration and Supply Agreement with Bayer to Evaluate OP-3136 in Combination with NUBEQA (darolutamide) in Metastatic Castration-Resistant Prostate Cancer | Study to evaluate OP-3136, Olema's novel KAT6 inhibitor, in combination with darolutamide in approximately 36 patients; expected to initiate in H2 2026First clinical collaboration for OP-3136; results... ► Artikel lesen | |
| DISC MEDICINE | 68,00 | 0,00 % | Disc Medicine launches expanded access program for bitopertin | ||
| FULCRUM THERAPEUTICS | 3,140 | 0,00 % | Fulcrum Therapeutics, Inc.: Fulcrum Therapeutics Announces Discontinuation of Pociredir Program in Sickle Cell Disease and Initiation of Strategic Review | ? Decision follows FDA feedback regarding the implications of the secondary malignancies observed with Tazverik- (tazemetostat) and the product's subsequent global withdrawal on the benefit-risk... ► Artikel lesen | |
| RECURSION PHARMACEUTICALS | 3,615 | +0,14 % | Recursion Pharmaceuticals: Recursion Reports Grant of Inducement Awards as Permitted by the Nasdaq Listing Rules | ||
| MINERALYS THERAPEUTICS | 28,850 | 0,00 % | Mineralys Therapeutics, Inc.: Mineralys Therapeutics Presents New Data from the Phase 3 Launch-HTN Trial of Lorundrostat in Participants with Hypertension and Chronic Kidney Disease at European Meeting on Hypertension and Cardiovascular Protection ... | - Post hoc analysis from pivotal Launch-HTN trial shows statistically significant and clinically meaningful reductions in blood pressure in participants with chronic kidney disease - - In participants... ► Artikel lesen | |
| CG ONCOLOGY | 55,47 | -0,11 % | CG Oncology, Inc. - 8-K, Current Report |
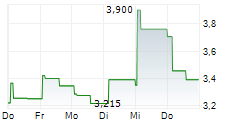
Das Unternehmen XUANZHU BIOPHARMACEUTICAL CO LTD kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell -4,99 % bei einem Aktienkurs von 1,712 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu XUANZHU BIOPHARMACEUTICAL CO LTD finden Sie auf Wallstreet Online.