- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 13.05. | ZyVersa Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Update | 266 | GlobeNewswire (Europe) | ZyVersa is advancing a highly differentiated pipeline focusing on inflammatory and renal diseases with a total accessible market >$100 billion.Inflammasome ASC Inhibitor IC 100 is the next evolution... ► Artikel lesen | |
| 13.05. | ZyVersa Therapeutics, Inc. - 10-Q, Quarterly Report | 4 | SEC Filings | ||
| 31.03. | ZyVersa Therapeutics GAAP EPS of -$4.18 | 1 | Seeking Alpha | ||
| ZYVERSA THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 31.03. | ZyVersa Therapeutics, Inc. - 10-K, Annual Report | - | SEC Filings | ||
| 05.03. | ZyVersa Therapeutics, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 19.11.25 | ZyVersa Therapeutics Reports Third Quarter 2025 Financial Results | 190 | GlobeNewswire (Europe) | ZyVersa is advancing a therapeutic development pipeline with multiple programs built around its two proprietary technologies - Cholesterol Efflux Mediator VAR 200 for treatment of kidney diseases... ► Artikel lesen | |
| 03.09.25 | ZyVersa Therapeutics Highlights Lipidomic Data in Alport Syndrome and DKD Reinforcing the Need for Drugs to Attenuate Damaging Renal Lipid Accumulation to Mitigate Disease Progression | 261 | GlobeNewswire (Europe) | ZyVersa is developing Cholesterol Efflux Mediator VAR 200 to mediate removal of damaging excess cholesterol and other lipids from the kidneys' filtration system. VAR 200 directly removes lipids from... ► Artikel lesen | |
| 13.08.25 | ZyVersa Therapeutics Reports Second Quarter 2025 Financial Results and Highlights Key Near-term Value-building Milestones | 525 | GlobeNewswire (Europe) | KEY HIGHLIGHTS First clinical site for Phase 2a clinical trial for Cholesterol Efflux Mediator VAR 200 in patients with diabetic kidney disease (DKD) was activated June 2025; patient screening... ► Artikel lesen | |
| 21.07.25 | ZyVersa Therapeutics CEO Issues Mid-year Shareholder Letter Highlighting Recent Corporate Developments and R&D Progress | 260 | GlobeNewswire (Europe) | WESTON, Fla., July 21, 2025 (GLOBE NEWSWIRE) -- ZyVersa Therapeutics, Inc. (Nasdaq: ZVSA; "ZyVersa"), a clinical stage specialty biopharmaceutical company developing first-in-class drugs for treatment... ► Artikel lesen | |
| 08.07.25 | ZyVersa Therapeutics Supports FDA-Authorized Emergency Compassionate Use of Cholesterol Efflux Mediator VAR 200 in a Patient with ApoCII Amyloidosis | 639 | GlobeNewswire (Europe) | ApoCII amyloidosis, resulting from abnormal organ deposition of toxic fibrillar amyloid proteins, is an ultra-rare condition that mostly affects the kidneys and manifests with protein spillage into... ► Artikel lesen | |
| 26.06.25 | ZyVersa Therapeutics Announces First Clinical Site Activation, Initiating Patient Recruitment for Cholesterol Efflux Mediator VAR 200's Phase 2a Clinical Trial in Patients with Diabetic Kidney Disease (DKD) | 269 | GlobeNewswire (Europe) | Initiation of patient recruitment marks a key milestone in the development of VAR 200, a potential first-in-class treatment for kidney disease addressing renal lipotoxicity.There are no available... ► Artikel lesen |
11 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| PRAXIS PRECISION MEDICINES | 254,00 | 0,00 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Vormatrigine Program Update | POWER1 Study in highly refractory patients with focal onset seizures did not meet its primary success measure Secondary measure, the 50% response rate, was met and seizure reduction during the second... ► Artikel lesen | |
| EDGEWISE THERAPEUTICS | 35,590 | 0,00 % | Servier to Acquire Edgewise Therapeutics' Muscular Dystrophy Business | Acquisition supports Servier's strategic ambition in rare neurology for patients with high unmet medical needs.Transaction includes sevasemten, a late-stage investigational treatment for... ► Artikel lesen | |
| JADE BIOSCIENCES | 16,940 | 0,00 % | Jade Biosciences Announces Positive Interim Phase 1 Results for JADE101 Demonstrating Potential Best-in-Class Profile and Every-12-Week Dosing in IgA Nephropathy | ~70% IgA reductions sustained at 12 weeks after a single dose of JADE101>70% IgA reductions projected at steady state with a single subcutaneous maintenance injection every 12 weeksIgA-lowering effect... ► Artikel lesen | |
| QIAGEN | 31,920 | -2,18 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Feedback aus Gesprächen mit etlichen US-Laborexperten deute darauf hin, dass... ► Artikel lesen | |
| APOGEE THERAPEUTICS | 86,86 | -0,07 % | Weekly Buzz: AstraZeneca, JNJ Get FDA Nod; Eli Lilly To Acquire Curevo; APGE Advances Zumilokibart | NEW BRUNSWICK (dpa-AFX) - This week in the biotech space, there were regulatory approvals across the U.S. and Europe, collaborations, acquisitions, and positive data readouts spanning multiple... ► Artikel lesen | |
| ADMA BIOLOGICS | 7,970 | 0,00 % | ADMA BIOLOGICS, INC. - 8-K, Current Report | ||
| SUMMIT THERAPEUTICS | 14,755 | -0,10 % | All You Need to Know About Summit Therapeutics (SMMT) Rating Upgrade to Buy | ||
| ERASCA | 11,970 | 0,00 % | Erasca, Inc.: Erasca Reports First Quarter 2026 Business Updates and Financial Results | Robust monotherapy efficacy and generally well-tolerated safety results observed during dose escalation for ERAS-0015 in both KRAS G12X NSCLC and PDAC reinforce best-in-class potential across RAS-targeted... ► Artikel lesen | |
| RECURSION PHARMACEUTICALS | 3,315 | 0,00 % | Recursion Pharmaceuticals: Recursion Reports First Quarter Financial Results and Provides Business Update | Multiple milestones achieved or on track across wholly owned and partnered programs REC-1245 (RBM39 degrader): Early clinical data in solid tumors demonstrate a well-tolerated safety profile and predictable... ► Artikel lesen | |
| MINERALYS THERAPEUTICS | 23,720 | +0,13 % | Mineralys Therapeutics, Inc.: Mineralys Therapeutics Announces Repurchase of Royalty Obligations in Tanabe License Agreement and Concurrent Financing | - Repurchase of lorundrostat royalty obligation in Tanabe license agreement for $200 million upfront and up to $100 million once certain commercial milestones are met - - $500 million committed... ► Artikel lesen | |
| TARSUS PHARMACEUTICALS | 59,28 | 0,00 % | Tarsus Pharmaceuticals, Inc: Tarsus Doses First Participant in Calliope, A Phase 2 Clinical Trial of TP-05 (lotilaner), a Novel Investigational Oral Tablet for the Potential Prevention of Lyme Disease | - A pioneering investigational oral prophylactic approach designed to kill Lyme-infected ticks before disease transmission - IRVINE, Calif., March 31, 2026 (GLOBE NEWSWIRE) -- Tarsus Pharmaceuticals... ► Artikel lesen | |
| RELAY THERAPEUTICS | 13,520 | 0,00 % | Relay Therapeutics Shares Jump 18% | ||
| BEAM THERAPEUTICS | 29,370 | -0,10 % | Beam Therapeutics Announces Compelling Updated Clinical Data from the Ongoing Phase 1/2 Trial of BEAM-302 in Alpha-1 Antitrypsin Deficiency (AATD) to Support Advancement to Pivotal Development | Treatment with 60 mg of BEAM-302 Led to Mean Steady-state Total AAT Level of 16.1 µM and All Patients Consistently and Durably Above the 11 µM Protective AAT Threshold with up to 12 Months of Follow-up... ► Artikel lesen | |
| TANGO THERAPEUTICS | 20,190 | -0,15 % | Tango Therapeutics, Inc.: Tango Therapeutics Announces Inducement Grants Under Nasdaq Listing Rule 5635(c)(4) | ||
| BIOCARDIA | 1,070 | +3,88 % | BioCardia Shares Surge 48% After FDA Feedback Supports Potential CardiAMP Approval Pathway |
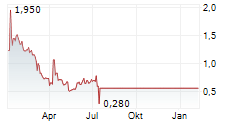
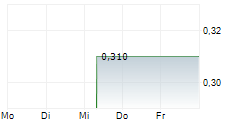
ZYVERSA THERAPEUTICS INC gehört der Branche Biotechnologie an. Der aktuelle Kurs liegt bei 0,310 Euro und damit -0,42 % im Minus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ZYVERSA THERAPEUTICS INC finden Sie auf Wallstreet Online.