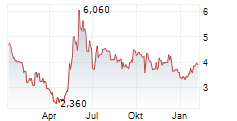
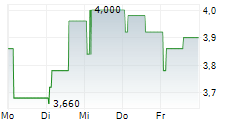
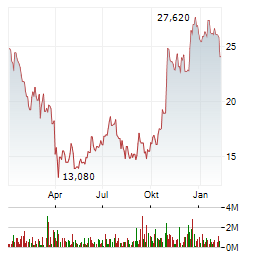
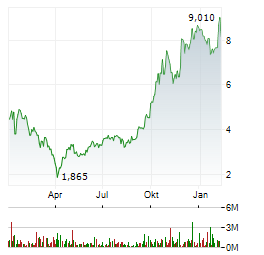
Chart-Typ:
Zeitraum:
| Immunovant Inc.: Immunovant Provides Corporate Updates and Reports Financial Results for the Fourth Quarter and Fiscal Year Ended March 31, 2026 | IMVT-1402 showed clinically meaningful response rates of 72.7% ACR20, 54.5% ACR50 and 35.8% ACR70 at Week 16 in the open label period of its trial in difficult-to-treat rheumatoid arthritis (D2T RA)... ► Artikel lesen |
| Immunovant Inc.: Immunovant Announces Phase 3 Study Results for Batoclimab in Thyroid Eye Disease (TED) | Phase 3 studies of batoclimab in thyroid eye disease (TED) each failed to meet their primary endpoint; safety results were consistent with previous findingsPatients in the TED studies demonstrated... ► Artikel lesen |
| Immunovant Inc.: Immunovant Provides Corporate Updates and Reports Financial Results for the Third Quarter Ended December 31, 2025 | IMVT-1402 potentially registrational trial in difficult-to-treat rheumatoid arthritis (D2T RA) fully enrolled, with topline data expected in the second half of calendar year 2026; topline data from... ► Artikel lesen |
| Relay Therapeutics, Inc.: Relay Therapeutics Announces Pricing of Public Offering of Common Stock | CAMBRIDGE, Mass., May 20, 2026 (GLOBE NEWSWIRE) -- Relay Therapeutics, Inc. (Nasdaq: RLAY), a clinical-stage, small molecule precision medicine company developing potentially life-changing therapies... ► Artikel lesen |
| Relay Therapeutics, Inc.: Relay Therapeutics Announces Initial Clinical Data Demonstrating That Zovegalisib Has Potential for Differentiated Safety and Efficacy in Patients with PIK3CA-Driven Vascular Anomalies | Promising initial efficacy data with 60% volumetric response rate across doses and 29%* at the lowest tested dose of 100mg twice daily (BID) with all patients ongoing Interim investigator- and patient-reported... ► Artikel lesen |
| Relay Therapeutics, Inc.: Relay Therapeutics Reports First Quarter 2026 Financial Results and Corporate Updates | FDA Breakthrough Therapy designation granted to zovegalisib for PIK3CA-mutant, HR+/HER2- advanced breast cancer, the Phase 3 ReDiscover-2 trial population in 2L breast cancer Presented zovegalisib... ► Artikel lesen |
| Tango Therapeutics, Inc.: Tango Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Highlights | First clinical data from PRMT5/RAS(ON) combination trial to be presented in 2026 Cash position of $380 million as of March 31, 2026, with runway into 2028 beyond anticipated key data inflection points... ► Artikel lesen |
| Tango Therapeutics Sub, Inc: Tango Therapeutics Announces Leadership Appointments to Accelerate Late-Stage Development of Vopimetostat | - Matthew Gall appointed Chief Financial Officer -- Additional key appointments add expertise in clinical operations, corporate strategy and project leadership - BOSTON, April 15, 2026 (GLOBE... ► Artikel lesen |
| Erasca, Inc.: Erasca and Tango Therapeutics Enter into Clinical Collaboration to Evaluate Combination of ERAS-0015 and Vopimetostat | ERAS-0015, a pan-RAS molecular glue, will be evaluated in combination with PRMT5 inhibitor vopimetostat Tango will sponsor the clinical trial and Erasca will supply ERAS-0015 at no cost SAN DIEGO... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu BIOSERGEN AB finden Sie auf Wallstreet Online.