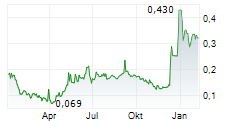
Nanexa announced today that all pharmacokinetic (PK) samples from the final dose group, 30 mg of the Phase I study for NEX-22, now have been analyzed. The results show increased exposure in line with the dose escalation and further demonstrate a controlled and prolonged release of liraglutide, which supports a one-month depot of liraglutide.
"The full results of the NEX-22 study will be presented at scientific congresses, with participation at the ADA (American Diabetes Association) in June being one of the highlights," says David Westberg, CEO of Nanexa. "The results also strengthen our position in our discussions with potential partners for the NEX-22 project."
For additional information, please contact:
David Westberg - CEO, Nanexa AB (publ)
Phone: +46 70 942 83 03
Email: david.westberg@nanexa.se
www.nanexa.com
The company's Certified Adviser is DNB Carnegie Investment Bank AB (publ).
About Nanexa AB (publ)
Nanexa is a pharmaceutical company developing injectable drug products based on the proprietary and innovative drug delivery system PharmaShell® - the high drug load delivery system enabling the next generation long-acting injectables through atomic layer precision. Nanexa develops its own products and also has collaboration agreements with several pharma companies, among others Novo Nordisk and AstraZeneca.
Nanexa's share is listed on Nasdaq First North Growth Market in Stockholm (NANEXA).