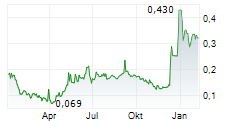
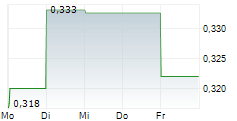
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 12.05. | Nanexa AB: Bulletin from the Annual General Meeting of Nanexa AB | 213 | GlobeNewswire (Europe) | The Annual General Meeting (AGM) of Nanexa AB (publ) has been held on May 12, 2026, at which meeting submitted proposals were passed. The complete proposals for resolutions are set out in the AGM notice.
The... ► Artikel lesen | |
| 29.04. | Nanexa AB: Nanexa publishes interim report for January-March 2026 | 366 | GlobeNewswire (Europe) | Opened the year with strong preclinical results for once-monthly and quarterly dosing of semaglutide
Significant events during the first quarter of 2026In January, Nanexa announced breakthrough preclinical... ► Artikel lesen | |
| 28.04. | Nanexa AB: Nanexa resolves on set-off of outstanding loan | 92 | GlobeNewswire (Europe) | NOT FOR RELEASE, PUBLICATION, OR DISTRIBUTION, DIRECTLY OR INDIRECTLY, IN OR INTO THE UNITED STATES, AUSTRALIA, BELARUS, JAPAN, CANADA, HONG KONG, NEW ZEALAND, SWITZERLAND, SINGAPORE, SOUTH AFRICA,... ► Artikel lesen | |
| 19.03. | Nanexa AB: Nanexa Demonstrates Feasibility of Quarterly Semaglutide Dosing with PharmaShell | 364 | GlobeNewswire (Europe) | Nanexa's atomic layer deposition (ALD)-based PharmaShell® platform enables an unprecedented pharmacokinetic profile for semaglutide, also supporting the potential for once-quarterly administration.New... ► Artikel lesen | |
| 19.02. | Nanexa AB: Nanexa publishes year-end report and Q4 report 2025 | 178 | GlobeNewswire (Europe) | The year ended strongly with an important milestone; the license and option agreement with Moderna, which is an important validation of our technology and has created favourable conditions for 2026... ► Artikel lesen | |
| 27.01. | Nanexa AB: Nanexa Announces Breakthrough Preclinical Data Demonstrating Exceptional Pharmacokinetic Profile for Monthly Semaglutide Formulation | 239 | GlobeNewswire (Europe) | Nanexa's atomic layer deposition (ALD) platform PharmaShell® demonstrably improves the pharmacokinetic profile of semaglutide injections.Significantly smoother plasma concentration curve could mitigate... ► Artikel lesen | |
| NANEXA Aktie jetzt für 0€ handeln | |||||
| 11.12.25 | Moderna signs deal worth up to $503m with Nanexa | 37 | Pharmaceutical Technology | ||
| 11.12.25 | Moderna taps Nanexa to improve delivery of injectable therapies in back-loaded $500M pact | 19 | FierceBiotech | ||
| 10.12.25 | 59 NORTH COMMUNICATIONS: Nanexa and Moderna enter into license and option agreement for the development of PharmaShell-based products | 15 | Cision News | ||
| 10.12.25 | Nanexa AB: Nanexa and Moderna enter into license and option agreement for the development of PharmaShell-based products | 447 | GlobeNewswire (Europe) | The agreement covers the development of up to five undisclosed compounds. Nanexa will receive an upfront payment of USD 3 million and is entitled to up to USD 500 million in potential milestone payments... ► Artikel lesen | |
| 06.11.25 | Nanexa AB: Nanexa publishes interim report for January-September 2025 | 253 | GlobeNewswire (Europe) | We have progressed with the optimization of our GLP-1 formulations, extended an existing commercial partnership, received approval for a PharmaShell patent application in Japan and submitted three new... ► Artikel lesen | |
| 22.09.25 | Nanexa AB: Nanexa receives Japanese patent approval for specific PharmaShell structure | 533 | GlobeNewswire (Europe) | The approval strengthens Nanexa's patent portfolio, with additional patent applications filed in other strategically important countries
Nanexa announces that a key patent application has been granted... ► Artikel lesen | |
| 27.08.25 | Nanexa AB: Nanexa publishes interim report for January-June 2025 | 271 | GlobeNewswire (Europe) | The second quarter of 2025 has continued to validate our core belief at Nanexa that slow-release medicines can transform how we dose and therefore treat people living with a range of conditions. Reducing... ► Artikel lesen |
13 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 33,460 | -4,75 % | Märkte am Morgen: Delivery Hero, MTU, Lufthansa, TUI, Airbus, Bayer, Space X | Nach der außergewöhnlichen Feiertagskonstellation - Pfingstmontag mit Handel in Deutschland, Memorial Day ohne Handel in den USA - tritt der DAX heute leicht auf die Bremse (-0,3 % erwartet). Gestern... ► Artikel lesen | |
| MERCK KGAA | 130,85 | 0,00 % | DAX-Check LIVE: Delivery Hero, Infineon, Merck, Nordex, Secunet, Siemens im Fokus | Der deutsche Aktienmarkt hat den Pfingstmontag mit einem fulminanten Kurssprung begangen. Der DAX startete mit Gewinnen, übersprang direkt die wichtige Marke von 25.000 Punkten und kletterte im Verlauf... ► Artikel lesen | |
| NOVO NORDISK | 37,750 | -1,05 % | Novo Nordisk-Aktie: Droht jetzt ein Kurssturz um -30%? | Der jüngste Aufwärtstrend der Novo Nordisk-Aktie ging Mitte Mai zu Ende. Der Kurs des dänischen Pharmakonzerns schaffte es nicht mehr über die Hürde von 300 DKK. Und nun droht nach Einschätzung eines... ► Artikel lesen | |
| PFIZER | 22,020 | -0,07 % | BARCLAYS stuft PFIZER INC auf 'Underweight' | LONDON (dpa-AFX Analyser) - Die britische Investmentbank Barclays hat die Einstufung für Pfizer mit einem Kursziel von 25 US-Dollar auf "Underweight" belassen. Angesichts der bisherigen Erfahrungen... ► Artikel lesen | |
| ELI LILLY | 929,40 | -0,10 % | HBM Healthcare Investments AG: Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden | HBM Healthcare Investments AG
/ Schlagwort(e): Ankauf
Eli Lilly übernimmt HBM-Portfoliounternehmen Curevo für bis zu USD 1.5 Milliarden
26.05.2026 / 13:10 CET/CEST
Die... ► Artikel lesen | |
| BRIGHT MINDS BIOSCIENCES | 81,47 | +0,56 % | Bright Minds Biosciences Announces Closing of US$175 Million Public Offering | NEW YORK, Jan. 09, 2026 (GLOBE NEWSWIRE) -- Bright Minds Biosciences Inc. (CSE: DRUG) (NASDAQ: DRUG) ("Bright Minds" or the "Company") today closed its recently announced public offering of 1,945... ► Artikel lesen | |
| ETON PHARMACEUTICALS | 28,680 | 0,00 % | Eton Pharmaceuticals Expands Rare Disease Portfolio Through Agreement for U.S. Commercialization Rights to IMPAVIDO (miltefosine) | IMPAVIDO is the first and only FDA-approved oral therapy for visceral, cutaneous, and mucosal leishmaniasis caused by specific Leishmania speciesLeishmaniasis is a rare but potentially life-threatening... ► Artikel lesen | |
| VERTEX PHARMACEUTICALS | 375,95 | -0,27 % | Vertex: FDA Accepts BLA Submission For Povetacicept; Assigns PDUFA Target Action Date Of Nov. 30 | CAMBRIDGE (MASSACHUSETTS) (dpa-AFX) - Vertex Pharmaceuticals Incorporated (VRTX) announced the FDA has accepted its Biologics License Application submission for povetacicept, an investigational... ► Artikel lesen | |
| GENERATE BIOMEDICINES | 12,970 | 0,00 % | Generate:Biomedicines: Generate Biomedicines, Inc. Reports First Quarter 2026 Financial Results and Provides Business Update | SOLAIRIA-1 AND SOLAIRIA-2 Phase 3 clinical trials for GB-0895 (anti-TSLP) continue to advance
Clinical trial sites activated for GB-4362 (MMAE neutralizer); first... ► Artikel lesen | |
| NOVARTIS | 125,94 | +0,27 % | DEUTSCHE BANK RESEARCH stuft NOVARTIS AG auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Novartis anlässlich der Investorenveranstaltung "dbAccess European Champions Conference" mit einem Kursziel von 135 Franken... ► Artikel lesen | |
| ABBOTT LABORATORIES | 75,46 | +0,05 % | EQS-News: Abbott erhält CE-Kennzeichnung für die weltweit erste duale Glukose-Keton-Sensortechnologie für Menschen mit Diabetes | EQS-News: Abbott
/ Schlagwort(e): Expansion/Sonstiges
Abbott erhält CE-Kennzeichnung für die weltweit erste duale Glukose-Keton-Sensortechnologie für Menschen mit Diabetes
27.05.2026... ► Artikel lesen | |
| ABBVIE | 183,30 | +0,11 % | AbbVie Announces European Commission Approval of AQUIPTA (atogepant) for the Acute Treatment of Migraine in Adults | AQUIPTA (atogepant) is now approved in the European Union for the acute treatment of migraine in adults, with or without aura, and for the prophylaxis of migraine in adults who have at least... ► Artikel lesen | |
| MERCK & CO | 98,68 | -0,35 % | Ihre wichtigsten Termine: Alle Analysten-Augen auf: Aroundtown, Kingfisher sowie Merck & Co | © Foto: © 2023 Merck & CoGut geplant in den Tag. Mit dem wO-Tagesausblick haben Sie die wichtigsten Termine im Blick und starten bestens vorbereitet in den neuen Handelstag.Unternehmenstermine 06:45... ► Artikel lesen | |
| GSK | 21,170 | -0,38 % | GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff | DJ GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff
Von Adria Calatayud
DOW JONES--GSK hat mit einem Medikamentenkandidaten gegen Hepatitis B einen teilweisen Therapieerfolg... ► Artikel lesen | |
| ZOETIS | 66,74 | +0,09 % | Argus downgrades Zoetis stock rating on competition, margin pressure |
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu NANEXA AB finden Sie auf Wallstreet Online.