WASHINGTON (dpa-AFX) - Viatris Inc. (VTRS), a healthcare company, on Wednesday said the U.S. Food and Drug Administration (FDA) has accepted its supplemental New Drug Application (sNDA) for phentolamine ophthalmic solution 0.75% for the treatment of presbyopia.
The regulator has set a PDUFA goal date of October 17, 2026.
Presbyopia is an age-related decline in the eye's ability to focus on nearby objects.
The submission is supported by data from the pivotal Phase 3 VEGA-2 and VEGA-3 trials, which met primary and all key secondary endpoints with no treatment-related serious adverse events reported.
Phentolamine ophthalmic solution 0.75% is currently approved in the U.S. for treating pharmacologically-induced mydriasis and is marketed as Ryzumvi.
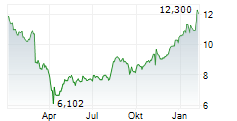
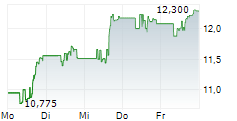
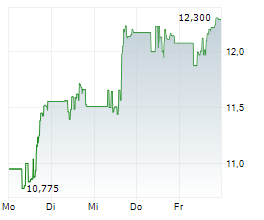
Viatris shares were up more than 1% in pre-market trading after closing at $16.07 on Tuesday.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News