PARIS (dpa-AFX) - Sanofi SA (SNY) on Friday said the European Medicines Agency's Committee for Medicinal Products for Human Use has issued a positive opinion for Acoziborole Winthrop for the treatment of both early and advanced-stage gambiense sleeping sickness in patients aged 12 years and older.
The recommendation was based on a Phase 2/3 study showing success rates of up to 96% at 18 months across both early and advanced stages of Trypanosoma brucei gambiense infection.
Human African trypanosomiasis, also known as sleeping sickness, is transmitted by the bite of an infected tsetse fly and is almost always fatal if left untreated.
Sanofi said it will donate Acoziborole Winthrop, co-developed with the Drugs for Neglected Diseases initiative (DNDi), to the World Health Organization through its philanthropic arm, Foundation S - The Sanofi Collective.
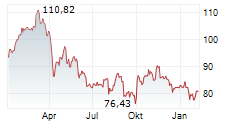
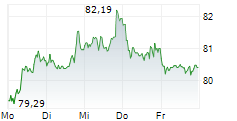
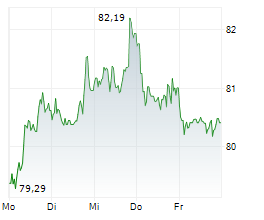
Sanofi shares closed at $48.35 on Thursday, up 1.60%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News