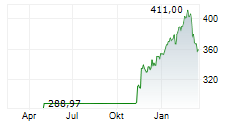
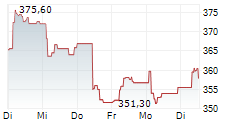
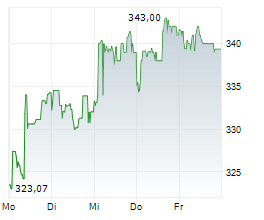
SOUTH SAN FRANCISCO (dpa-AFX) - Roche Holding AG (RHHBY) on Monday said its pivotal Phase III FENhance 1 study of fenebrutinib in relapsing multiple sclerosis (RMS) met its primary endpoint, demonstrating a significant reduction in disease relapse rates.
Multiple sclerosis is a chronic autoimmune disease in which the immune system attacks the protective covering of nerves in the brain and spinal cord, disrupting communication between the brain and body and causing symptoms such as vision loss, fatigue, numbness and mobility problems.
The study showed that fenebrutinib reduced the annualised relapse rate (ARR) by 51% compared with Sanofi's teriflunomide over at least 96 weeks of treatment. This was consistent with results from the FENhance 2 trial, which showed a 59% reduction in ARR. Together, the findings equate to approximately one relapse every 17 years.
Roche added that full data from the FENhance 1 and 2 studies will be shared at the American Academy of Neurology (AAN) Annual Meeting 2026 and submitted to regulatory authorities.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News