PARIS (dpa-AFX) - Sanofi (SNY, SAN.PA) announced the Ministry of Health, Labour and Welfare in Japan has granted orphan drug designation to rilzabrutinib, an oral, reversible covalent Bruton's tyrosine kinase inhibitor, for IgG4-related disease. Currently, rilzabrutinib in IgG4-RD is being evaluated in the RILIEF phase 3 study.
Rilzabrutinib is being studied across multiple rare immune-mediated diseases. In 2025, it received approval for immune thrombocytopenia in the US, the EU, and the UAE. Additionally, rilzabrutinib is currently under regulatory review for ITP in Japan.
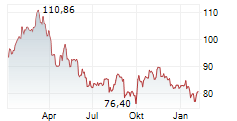
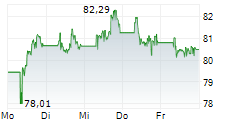
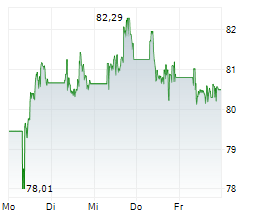
At last close, Sanofi was trading at 82.20 euros.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News