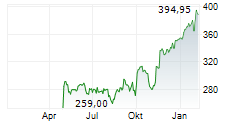
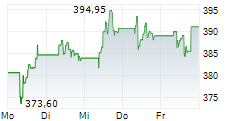
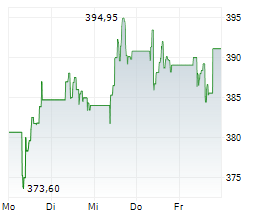
SOUTH SAN FRANCISCO (dpa-AFX) - Genentech, part of the Roche Holding AG (RHHBY), on Friday said a detailed analysis from the Phase 3 ALLEGORY Trial of Gazyva in adults with Systemic Lupus Erythematosus was published in the New England Journal of Medicine.
The study met its primary endpoint, with 76.7% of patients receiving Gazyva plus standard therapy achieving at least a four-point improvement in the SLE Responder Index-4 at 52 weeks, compared with 53.5% for placebo plus standard therapy.
The treatment also showed superiority across key secondary endpoints, including longer time to first disease flare and higher remission rates compared with placebo. Results are also being presented at the European Lupus Meeting SLEuro 2026.
The company said the data are being discussed with regulators, including the U.S. Food and Drug Administration and the European Medicines Agency.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News