WASHINGTON (dpa-AFX) - Incyte (INCY) announced that the European Commission has approved Zynyz (retifanlimab) in combination with carboplatin and paclitaxel, a platinum-based chemotherapy regimen, for the first-line treatment of adult patients with metastatic or inoperable locally recurrent squamous cell carcinoma of the anal canal (SCAC).
This decision follows the positive opinion issued in January 2026 by the European Medicines Agency's Committee for Medicinal Products for Human Use (CHMP). It marks the second indication for Zynyz in Europe, which was previously approved as a monotherapy for the first-line treatment of adult patients with metastatic or recurrent locally advanced Merkel cell carcinoma (MCC).
The approval is supported by data from the Phase 3 POD1UM-303/InterAACT2 trial, which evaluated Zynyz or placebo in combination with carboplatin and paclitaxel in adult patients with metastatic or inoperable locally recurrent SCAC who had not previously received systemic chemotherapy.
Zynyz is also indicated as monotherapy for the first-line treatment of adult patients with metastatic or recurrent locally advanced MCC in the U.S., EU, Canada, and Switzerland. Incyte markets Zynyz in the United States and, since 2017, has held global rights to retifanlimab through an exclusive collaboration and license agreement with MacroGenics, Inc.
For More Such Health News, visit rttnews.com.
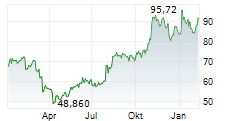
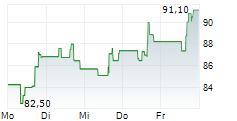
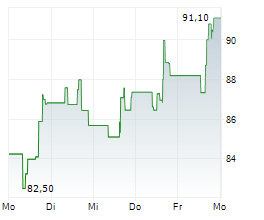
INCY ended trading on March 6 at $95.94, down $1.39 or 1.43%. In after-hours trading, the stock slipped further to $95.40, a decline of $0.54 or 0.56%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News