SOUTH SAN FRANCISCO (dpa-AFX) - Genentech, a member of the Roche Group (RO.SW, ROG.SW), announced results from the Phase III persevERA Breast Cancer study evaluating investigational giredestrant in combination with palbociclib for people with estrogen receptor-positive, human epidermal growth factor receptor 2-negative, locally advanced or metastatic breast cancer. The company stated that the study did not meet its primary objective of a statistically significant improvement in progression-free survival in the intent-to-treat population versus letrozole plus palbociclib, but a numerical improvement was observed.
The FDA recently accepted the NDA based on the evERA data. In the coming weeks, the company will submit the giredestrant Phase III lidERA data in early-stage breast cancer to the FDA.
Levi Garraway, chief medical officer and head of Global Product Development, said: 'We believe there is a path forward for combining giredestrant with a CDK4/6 inhibitor in the adjuvant setting and we are conducting further studies. The efficacy demonstrated in evERA and lidERA provides clear validation of the clinical activity of giredestrant and reinforces the strength of our expanding clinical development program.'
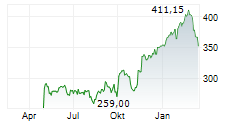
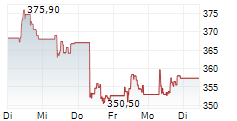
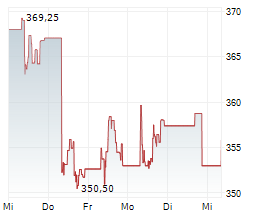
At last close, Roche shares were trading at 341.20 francs, down 2.9%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News