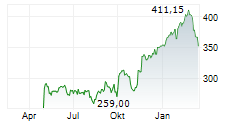
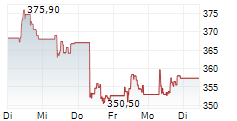
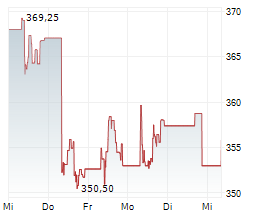
SOUTH SAN FRANCISCO (dpa-AFX) - Roche Holding AG (RHHBY) said on Monday it has received CE Mark approval for its Elecsys Apolipoprotein E4 biomarker test- the first in-vitro diagnostic (IVD) immunoassay to identify the presence of the ApoE4 gene variant in the bloodstream.
The ApoE4 gene variant is associated with an increased risk of late-onset Alzheimer's disease and is present in around 40-60% of patients with the condition.
The clinical performance of the assay was evaluated in participants with cognitive complaints or objective memory impairment of unknown cause. Results from the Elecsys ApoE4 test were compared with APOE4 genotyping performed using Sanger sequencing. The assay showed 100% concordance between ApoE4 protein detection and genetic APOE4 status, correctly identifying all genetic carriers and non-carriers.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News