NEW YORK CITY (dpa-AFX) - Pfizer Inc. (PFE) on Tuesday reported positive topline results from the Phase 2 FOURLIGHT-1 study evaluating atirmociclib in combination with fulvestrant in people with hormone receptor (HR)-positive, human epidermal growth factor receptor 2-negative (HER2-) advanced or metastatic breast cancer.
The study met its primary endpoint, showing a statistically significant and clinically meaningful improvement in progression-free survival.
FOURLIGHT-1 evaluated atirmociclib plus fulvestrant compared with fulvestrant alone or everolimus in combination with exemestane in adults receiving second-line treatment for advanced or metastatic breast cancer.
The progression-free survival benefit was consistent across all prespecified subgroups, including patients with differing performance status, menopausal status, presence of visceral disease, and prior exposure to CDK4/6 inhibitors.
'The strength of these data reinforces our confidence that atirmociclib may meaningfully differentiate from the CDK4/6 inhibitor class, the standard-of-care backbone in HR-positive breast cancer, with the potential for improved efficacy and tolerability,' said Jeff Legos, Chief Oncology Officer, Pfizer.
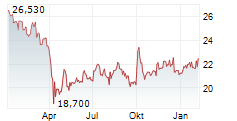
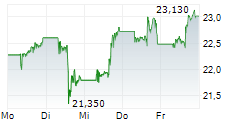
On Monday, Pfizer shares closed at $26.61, up 0.11%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News