PARIS (dpa-AFX) - Regeneron Pharmaceuticals (REGN) and Sanofi (SNY, SAN.PA) announced that the Ministry of Health, Labour and Welfare in Japan has approved Dupixent or dupilumab for the treatment of adults with moderate-to-severe bullous pemphigoid. The approval in Japan is based on data from the LIBERTY-BP-ADEPT Phase 2/3 trial.
Bullous pemphigoid is the seventh approved indication for Dupixent in Japan. It is approved in certain patients with atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyposis, prurigo nodularis, chronic spontaneous urticaria and chronic obstructive pulmonary disease.
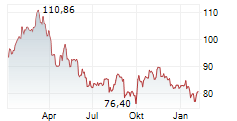
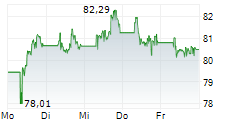
At last close, Sanofi shares were down 1.23% to 76.83 euros.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News