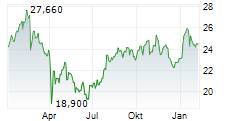
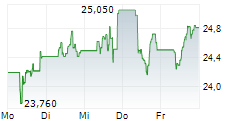
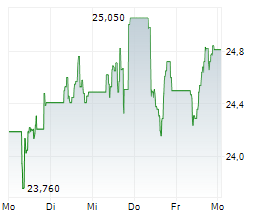
AMSTERDAM (dpa-AFX) - Medical technology company Royal Philips (PHG) announced Thursday it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for EchoNavigator R5.0 with DeviceGuide, an AI-powered software solution.
DeviceGuide assists physicians during one of interventional cardiology's most technically demanding procedures of repairing leaking mitral valves through a minimally invasive approach. Philips It uses AI to track and visualize mitral valve repair devices in real time during minimally invasive procedures, supporting treatment.
DeviceGuide was developed in close collaboration with Edwards Lifesciences Corp. (EW), the global leader in structural heart innovation, aligning Philips' imaging and AI expertise with Edwards' expertise in valvular heart therapy development.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News