PARIS (dpa-AFX) - Sanofi (SNY, SAN.PA) announced the European Medicines Agencys Committee for Medicinal Products for Human Use has adopted a positive opinion recommending the approval of Sarclisa or isatuximab subcutaneous in combination with approved standard-of-care regimens for the treatment of patients with multiple myeloma across all currently approved indications for Sarclisa intravenous formulation in the EU.
The company said if approved, Sarclisa subcutaneous would be the first available anticancer treatment to be administered through an on-body injector, and the first multiple myeloma medicine available by both SC OBI and manual injection in the EU.
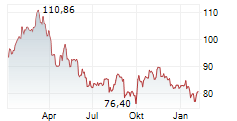
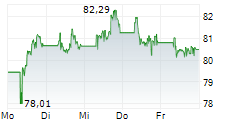
Sanofi shares are trading at 82.40 euros, up 1.54%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News