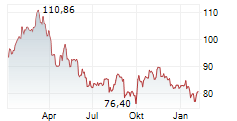
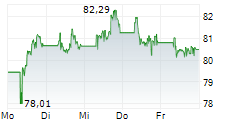
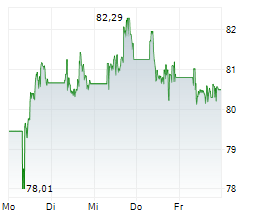
PARIS (dpa-AFX) - Sanofi (SNY, SAN.PA) said phase 2 studies of lunsekimig in two chronic respiratory diseases met their primary and key secondary endpoints compared to placebo. In both studies, lunsekimig was well tolerated, with an acceptable safety profile. Detailed results from the AIRCULES, DUET, and VELVET studies will be presented at upcoming medical congresses.
The AIRCULES phase 2b study achieved its primary and key secondary endpoints in moderate-to-severe asthma regardless of biomarker status. The DUET phase 2a study met its primary and key secondary endpoints in chronic rhinosinusitis with nasal polyps. The exploratory VELVET phase 2b study did not meet its primary endpoint in moderate-to-severe atopic dermatitis.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News