WESTON (dpa-AFX) - Thursday, Biogen Inc. (BIIB) announced compelling topline results from the Phase 2 CELIA study evaluating diranersen in participants with early Alzheimer's disease.
Notably, the study did not meet its primary endpoint assessing dose response for change from baseline on the Clinical Dementia Rating-Sum of Boxes at Week 76.
However, these results provide the first evidence from a randomized Phase 2 study of a tau-directed therapy demonstrating both robust biomarker impact and cognitive benefit in early Alzheimer's disease.
The company stated that the pre-specified analyses of cognitive endpoints showed slowing of clinical decline across all studied doses, particularly in participants receiving the lowest dose of diranersen, 60 mg administered every 24 weeks.
Diranersen also demonstrated robust reductions in both cerebrospinal fluid tau and tau pathology across all studied doses, with reductions maintained throughout the dosing period.
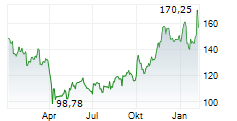
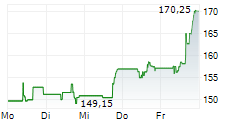
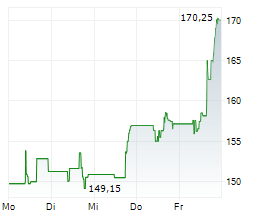
In the pre-market hours, BIIB is trading at $216.11, up 5.66 percent on the Nasdaq.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News