- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 14.05. | Biofrontera Inc. (BFRI) Reports Q1 Loss, Beats Revenue Estimates | 17 | Zacks | ||
| BIOFRONTERA INC Aktie jetzt für 0€ handeln | |||||
| 14.05. | Biofrontera Inc. - 8-K, Current Report | 2 | SEC Filings | ||
| 14.05. | Biofrontera Inc. Reports First Quarter 2026 Financial Results and Provides a Business Update | 366 | GlobeNewswire (Europe) | Woburn, MA, May 14, 2026 (GLOBE NEWSWIRE) -- Biofrontera Inc. (NASDAQ: BFRI) (the "Company"), a biopharmaceutical company specializing in the development and commercialization of photodynamic therapy... ► Artikel lesen | |
| 13.05. | Biofrontera Inc.: Biofrontera Launches Second Annual "Hats On" Campaign to Raise Awareness of Actinic Keratosis | 3 | GlobeNewswire (USA) | ||
| 19.03. | Biofrontera Inc. - 10-K, Annual Report | 8 | SEC Filings | ||
| 19.03. | Biofrontera Inc. - 8-K, Current Report | 6 | SEC Filings | ||
| 19.03. | Biofrontera Inc. Reports Record Fourth Quarter and Full Year 2025 Financial Results and Provides a Business Update | 339 | GlobeNewswire (Europe) | Conference call will be held today, Thursday, March 19 at 10:00 am ET Woburn, MA, March 19, 2026 (GLOBE NEWSWIRE) -- Biofrontera Inc. (NASDAQ:BFRI) (the "Company"), a biopharmaceutical company specializing... ► Artikel lesen | |
| 09.03. | Biofrontera Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 09.03. | Biofrontera Inc. Reports Positive Phase 2b Results Supporting Further Development of Ameluz Photodynamic Therapy for moderate to severe Acne Vulgaris | 359 | GlobeNewswire (Europe) | • Phase 2b study demonstrated greater reductions in inflammatory acne lesions with Ameluz, PDT versus vehicle • 3-hour incubation regimen identified as the most promising protocol for further clinical... ► Artikel lesen | |
| 26.02. | Biofrontera Inc. - 8-K, Current Report | 2 | SEC Filings | ||
| 26.02. | Biofrontera Inc.: U.S. Patent Office Issues Final Written Decision Finding All Challenged Claims of Sun Pharma's Patent 11,697,028 To Be Unpatentable | 443 | GlobeNewswire (Europe) | WOBURN, Mass., Feb. 26, 2026 (GLOBE NEWSWIRE) -- Biofrontera Inc. (Nasdaq: BFRI), a biopharmaceutical company specializing in the commercialization and development of photodynamic therapy ("PDT")... ► Artikel lesen | |
| 17.02. | Biofrontera Inc. Announces Database Lock of Phase 1 Pharmacokinetics Study of Ameluz for Actinic Keratoses on Trunk and Extremities | 302 | GlobeNewswire (Europe) | Phase 1 maximal-use pharmacokinetics (PK) study completed in support of planned U.S. label expansionData to complement previously announced positive Phase 3 efficacy results on extremities, neck and... ► Artikel lesen | |
| 11.02. | Biofrontera Inc.: Biofrontera Announces FDA Filing Acceptance of Supplemental New Drug Application for Ameluz PDT in Superficial Basal Cell Carcinoma | 405 | GlobeNewswire (Europe) | Prescription Drug User Fee Act (PDUFA) target action date set for September 28, 2026If approved, Ameluz would be the first and only PDT photosensitizer indicated for the treatment of superficial Basal... ► Artikel lesen | |
| 09.02. | Biofrontera Inc. - 8-K, Current Report | 3 | SEC Filings | ||
| 09.02. | Biofrontera Inc.: Biofrontera Announces Positive Results in Phase 3 Study of Ameluz PDT for Actinic Keratoses on the Extremities, Neck, and Trunk, Meeting Primary Endpoint | 382 | GlobeNewswire (Europe) | Study findings met its primary endpoint and showed highly statistically significant superiority for Ameluz vs. vehicle gel (pActinic keratosis (AK) is the most common skin condition diagnosed by US... ► Artikel lesen | |
| 08.01. | Biofrontera Inc. Announces Data Base Locks for Two Clinical Studies Supporting Key Data and Regulatory Milestones | 14 | GlobeNewswire (USA) | ||
| 07.01. | Biofrontera Inc. - 8-K, Current Report | 11 | SEC Filings | ||
| 18.12.25 | Biofrontera Inc. Completes Transfer of Ameluz and RhodoLED FDA approval and Associated Intellectual Property Portfolio | 358 | GlobeNewswire (Europe) | • Ameluz- and RhodoLED- New Drug Application (NDA) and Investigational New Drug Application (IND) have successfully been transferred to Biofrontera Inc.• Assignment to Biofrontera Inc. of 11 granted... ► Artikel lesen | |
| 04.12.25 | Biofrontera Inc.: Last Patient Completes Biofrontera's Phase 1 Pharmacokinetics Study of Ameluz for Treatment of Actinic Keratoses on the Trunk and Extremities | 345 | GlobeNewswire (Europe) | Phase 1 Maximal-use Phase 1 study evaluated pharmacokinetic (PK) profile following application of 3 tubes of Ameluz- over treatment area of 240 cm2Completion of this study marks a key milestone towards... ► Artikel lesen | |
| 13.11.25 | Biofrontera Inc. Reports Third Quarter 2025 Financial Results and Provides a Business Update | 590 | GlobeNewswire (Europe) | Conference call will be held today, Thursday, November 13 at 10:00 am ET Woburn, MA, Nov. 13, 2025 (GLOBE NEWSWIRE) -- Biofrontera Inc. (NASDAQ:BFRI) (the "Company"), a biopharmaceutical company specializing... ► Artikel lesen |
1 2 Weiter >>
36 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 36,160 | +1,69 % | Börsenrotation ohne KI! Jetzt 100 % mit MustGrow, Bayer und Novo Nordisk, Achtung bei K+S | Erstellt und veröffentlicht im Auftrag von MustGrow Biologics Corp. Perspektivenwechsel bleibt das Gebot der Stunde. Denn gestern tagte die EZB zur aktuellen Lage bei Inflation und Zinsniveau. Mit einer... ► Artikel lesen | |
| AURORA CANNABIS | 2,900 | -1,16 % | Aurora Deepens its Impact for Veterans Across Canada | ||
| DERMAPHARM | 47,550 | +0,21 % | wO Heimspiel: Nischenpharma mit 43-%-Marge: Das Dermapharm-Dilemma für Investoren | © Foto: Rolf-Peter Stoffels/Geisler-Foto - Geisler-FotopressDermapharm produziert Markenarzneimittel ohne Patentschutz und verdient dabei gut. Doch wie viel ist das wert? Drei Analysehäuser kommen zu... ► Artikel lesen | |
| MPH HEALTH CARE | 26,600 | +0,76 % | Original-Research: MPH Health Care AG (von First Berlin Equity Research GmbH): Kaufen | Original-Research: MPH Health Care AG - from First Berlin Equity Research GmbH
05.06.2026 / 12:45 CET/CEST
Dissemination of a Research, transmitted by EQS News - a service of EQS Group.
The issuer... ► Artikel lesen | |
| ARZNEIWERK AG VIDA | 0,262 | +0,77 % | PTA-AFR: Arzneiwerk AG VIDA: Vorabbekanntmachung über die Veröffentlichung von Finanzberichten gemäß §§ 114-117 WpHG | DJ PTA-AFR: Arzneiwerk AG VIDA: Vorabbekanntmachung über die Veröffentlichung von Finanzberichten gemäß §§ 114-117 WpHG
Vorabbekanntmachung Finanzberichte gemäß ---- 114-117 WpHG
Arzneiwerk... ► Artikel lesen | |
| SYNBIOTIC | 1,662 | -3,03 % | EQS-News: SYNBIOTIC SE: SYNBIOTIC startet mit operativem Fortschritt, neuen regulatorischen Impulsen und starker Branchenpräsenz ins Frühjahr 2026 | EQS-News: SYNBIOTIC SE
/ Schlagwort(e): Marktbericht/Prognose
SYNBIOTIC startet mit operativem Fortschritt, neuen regulatorischen Impulsen und starker Branchenpräsenz ins Frühjahr... ► Artikel lesen | |
| DIGICANN VENTURES | 0,003 | +25,00 % | Digicann Ventures Inc.: Digicann Ventures Announces Proposed Reverse Takeover Transaction with Winning Asia Technology Macau Limited | Vancouver, British Columbia--(Newsfile Corp. - June 5, 2026) - Digicann Ventures Inc. (CSE: DCNN.X) ("Digicann" or the "Company"), is a reporting issuer listed on the Canadian Securities Exchange... ► Artikel lesen | |
| SCHOTT PHARMA | 17,440 | -1,36 % | SCHOTT Pharma Expands Vial Production in Lebanon, PA | ||
| CANTOURAGE GROUP | 6,340 | -1,86 % | EQS-DD: Cantourage Group SE: Philip Schetter, Annahme der Gewährung von 25.000 Aktienoptionen (Bezugsrechten auf Aktien der Cantourage Group SE; ISIN: DE000A3DSV01) im Rahmen des von der ... | Meldung und öffentliche Bekanntgabe der Geschäfte von Personen, die Führungsaufgaben wahrnehmen, sowie in enger Beziehung zu ihnen stehenden Personen
05.06.2026 / 21:06... ► Artikel lesen | |
| COSMO | 77,90 | -1,02 % | Cosmo Pharmaceuticals N.V.: Cosmo Announces Completion of Phase II Enrolment in Distal Ulcerative Colitis Program | Dublin, Ireland--(Newsfile Corp. - May 18, 2026) - Cosmo N.V. (SIX: COPN) (XETRA: C43) ("Cosmo" or "the Company") today announced the completion of patient enrolment in its Phase II clinical... ► Artikel lesen | |
| SWEET EARTH | 0,324 | -100,00 % | Quantum-Aktie vor nächstem Schritt? Quantum X Labs integriert Google-Technologie - Markt für Quantencomputing gewinnt an Dynamik | ||
| RESTART LIFE SCIENCES | 0,054 | 0,00 % | Restart Life Sciences Corp.: Restart Life Sciences Appoints Inez Ho as Chief Financial Officer | Vancouver, British Columbia--(Newsfile Corp. - June 3, 2026) - Restart Life Sciences Corp. (CSE: HEAL) (FSE: HN30) (OTC Pink: NMLSF) ("Restart Life" or the "Company") is pleased to announce the... ► Artikel lesen | |
| AEQUUS PHARMACEUTICALS | 0,005 | -100,00 % | Aequus Pharmaceuticals Inc: Aequus closes sale of Zimed PF to Luvo for $531,693 | ||
| MERCK KGAA | 136,85 | -1,90 % | JPMORGAN stuft MERCK KGAA auf 'Overweight' | NEW YORK (dpa-AFX Analyser) - Die US-Bank JPMorgan hat die Einstufung für Merck KGaA mit einem Kursziel von 150 Euro auf "Overweight" belassen. Die Studiendaten von Varegacestat hätten zwar auf eine... ► Artikel lesen | |
| NOVO NORDISK | 37,860 | -0,56 % | Was bei Novo nordisk gerade passiert: Was die wenigsten Anleger auf dem Schirm haben - Bereiten Sie sich auf Montag vor |
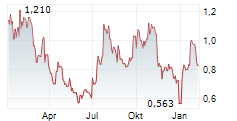
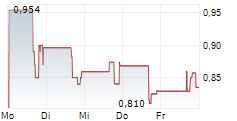
Das Unternehmen BIOFRONTERA INC kann der Branche Pharma zugeordnet werden. Mit einer aktuellen Kursveränderung von -0,004 (-0,37 %) liegt der Kurs bei 0,932 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu BIOFRONTERA INC finden Sie auf Wallstreet Online.