- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Mi | Longeveron Issues Letter to Stockholders Highlighting Corporate Strategy, Strategic Partnering Approach and 2026 Key Priorities | 46 | GlobeNewswire (Europe) | MIAMI, May 20, 2026 (GLOBE NEWSWIRE) -- Longeveron Inc. (NASDAQ: LGVN), a clinical stage biotechnology company developing cellular therapy for life-threatening, rare pediatric and chronic aging-related... ► Artikel lesen | |
| 14.05. | Longeveron anticipates August 2026 ELPIS II readout while shifting to an asset-light partnering model | 2 | Seeking Alpha | ||
| 13.05. | Longeveron Q1 2026 Earnings Call: Complete Transcript | 1 | Benzinga.com | ||
| 13.05. | Longeveron GAAP EPS of -$0.19 misses by $0.02, revenue of $0.4M beats by $0.08M | 1 | Seeking Alpha | ||
| 13.05. | Longeveron Inc. - 10-Q, Quarterly Report | 1 | SEC Filings | ||
| 11.05. | Longeveron Announces Independent Data Monitoring Committee Positive Review and Recommendation to Complete Phase 2b Stem Cell Therapy Clinical Trial in Hypoplastic Left Heart Syndrome (HLHS) | 5 | GlobeNewswire (USA) | ||
| 08.05. | Longeveron erleidet Rückschlag: FDA stuft HLHS-Studie nicht mehr als zulassungsrelevant ein | 2 | Investing.com Deutsch | ||
| LONGEVERON Aktie jetzt für 0€ handeln | |||||
| 08.05. | Longeveron faces setback as FDA no longer views HLHS trial as pivotal | 1 | Investing.com | ||
| 08.05. | Longeveron Announces Constructive Type C Meeting with U.S. FDA Ahead of Data Readout for ELPIS II Phase 2b Clinical Trial Evaluating Treatment for Hypoplastic Left Heart Syndrome (HLHS) | 270 | GlobeNewswire (Europe) | MIAMI, May 08, 2026 (GLOBE NEWSWIRE) -- Longeveron Inc. (NASDAQ: LGVN), a clinical stage regenerative medicine biotechnology company developing cellular therapies for life-threatening and chronic... ► Artikel lesen | |
| 08.05. | Longeveron Inc. - 8-K, Current Report | - | SEC Filings | ||
| 04.05. | H.C. Wainwright cuts Longeveron stock price target on dilution | 2 | Investing.com | ||
| 10.04. | Longeveron Inc. - 8-K, Current Report | - | SEC Filings | ||
| 10.04. | Longeveron Inc. - S-1, General form for registration of securities | - | SEC Filings | ||
| 08.04. | Longeveron receives China patent for stem cell potency assay | 5 | Investing.com | ||
| 08.04. | Longeveron erhält chinesisches Patent für Testverfahren von Stammzellen | 1 | Investing.com Deutsch | ||
| 08.04. | Longeveron Granted Chinese Patent for Potency Assay Methods for Assessing Human Mesenchymal Stem Cells (MSCs) | 1 | GlobeNewswire (USA) | ||
| 24.03. | Longeveron Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 17.03. | Longeveron Announces 2025 Full Year Financial Results and Provides Business Update | 1.035 | GlobeNewswire (Europe) | On track for third quarter 2026 top-line trial results from anticipated pivotal Phase 2b clinical trial (ELPIS II) evaluating laromestrocel as a potential adjunct treatment for HLHS, a rare pediatric... ► Artikel lesen | |
| 17.03. | Longeveron Inc. - 10-K, Annual Report | 1 | SEC Filings | ||
| 12.03. | Longeveron Inc. - 8-K, Current Report | 2 | SEC Filings |
1 2 3 Weiter >>
49 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| CRISPR THERAPEUTICS | 42,910 | +2,75 % | CRISPR Therapeutics AG: CRISPR Therapeutics Provides Business Update and Reports First Quarter 2026 Financial Results | ZUG, Switzerland and BOSTON, May 04, 2026 (GLOBE NEWSWIRE) -- CRISPR Therapeutics (Nasdaq: CRSP) today reported financial results for the first quarter ended March 31, 2026. "The first quarter reflected... ► Artikel lesen | |
| OCUGEN | 1,184 | +0,85 % | Ocugen schließt Platzierung von Wandelanleihen über 130 Millionen US-Dollar ab | ||
| EDITAS MEDICINE | 2,320 | +3,57 % | Editas Medicine, Inc.: Editas Medicine Reports New Preclinical Data Demonstrating Progress of EDIT-401 as Potential Treatment for Hyperlipidemia at the American Society of Gene and Cell Therapy 2026 Annual Meeting | CAMBRIDGE, Mass., May 14, 2026 (GLOBE NEWSWIRE) -- Editas Medicine, Inc. (Nasdaq: EDIT), a pioneering gene editing company developing transformative medicines for serious diseases, shared new preclinical... ► Artikel lesen | |
| PACIFIC BIOSCIENCES OF CALIFORNIA | 1,097 | +6,82 % | PacBio and Lucid Genomics Announce Compatibility Collaboration to Advance Tertiary Analysis for Long-Read Sequencing | MENLO PARK, Calif. & BERLIN, April 21, 2026 (GLOBE NEWSWIRE) -- PacBio (NASDAQ: PACB), a premier developer of sequencing solutions, and Lucid Genomics GmbH, a provider of cutting-edge bioinformatics... ► Artikel lesen | |
| DENALI THERAPEUTICS | 16,445 | +1,58 % | Denali Therapeutics: Erster kommerzieller Durchbruch der Blut-Hirn-Schranken-Plattform entfacht neue Fantasie - reicht das für Anleger? | Die Aktie von Denali Therapeutics steht nach der ersten kommerziellen Validierung ihrer Blut-Hirn-Schranken-Plattform im Fokus. Ein gemeinsames Produkt mit Biogen hat in der EU eine Zulassungsempfehlung... ► Artikel lesen | |
| SOL GLOBAL INVESTMENTS | 0,026 | -100,00 % | CSE Bulletin: Suspension - SOL Global Investments Corp. (SOL) | Le 7 avril/April 2026
Effective immediately, SOL Global Investments Corp. is suspended pursuant to CSE Policy 3. The suspension is considered a Regulatory Halt as defined in National Instrument 23-101... ► Artikel lesen | |
| FATE THERAPEUTICS | 1,777 | +12,54 % | Globaler Biotech-Wettlauf und die Zukunft der Plattformstrategie - Warum Mesoblast auf den Pfaden von großen Konzernen wie Merck KGaA wandert | ||
| TRINITY BIOTECH | 0,625 | +10,62 % | Trinity Biotech plc: Trinity Biotech Appoints Jerry Lydon to Lead North America Commercial Operations | - Appointment supports revenue quality and profitability momentum as the Company activates its transformed operating base - Company announces further operational progress in its Comprehensive Transformation... ► Artikel lesen | |
| VOYAGER THERAPEUTICS | 3,104 | +2,99 % | Voyager Therapeutics, Inc.: Voyager ASGCT Late Breaker: Single IV Dose of VY1706 Well Tolerated, Reduced Tau in 3-Month GLP Toxicology Data; Clinical Trial in Alzheimer's Disease Expected H2 2026 | - VY1706 IND application process on track for Q2 2026; clinical entry expected H2 2026 - - Data from eight ASGCT presentations highlight Voyager's continued innovation in gene therapy - LEXINGTON... ► Artikel lesen | |
| ARCTURUS THERAPEUTICS | 7,005 | +4,71 % | Arcturus outlines 12-week ARCT-032 Phase II enrollment and extends cash runway beyond Q2 2028 | ||
| ANAVEX LIFE SCIENCES | 2,244 | +2,89 % | ANAVEX LIFE SCIENCES CORP: Ruhe vor der nächsten Phase | ||
| BIOAGE LABS | 14,300 | +3,32 % | BioAge Labs, Inc.: BioAge Labs Reports First Quarter 2026 Financial Results and Provides Business Updates | Positive topline Phase 1 data for BGE-102, demonstrating potential best-in-class reductions among oral NLRP3 inhibitors in inflammatory biomarkers of cardiovascular risk Phase 2 dose-ranging proof-of-concept... ► Artikel lesen | |
| CERUS | 2,360 | +1,72 % | Cerus Corporation Announces New, Four-Year Supply Agreement with French Blood Establishment for INTERCEPT Blood System | Cerus Corporation (Nasdaq: CERS) announced today the signing of a new, four-year supply agreement with Établissement Français du Sang (EFS), the French Blood Establishment, for the INTERCEPT Blood... ► Artikel lesen | |
| LINEAGE CELL THERAPEUTICS | 1,062 | -3,98 % | Regenerative Medizin im Aufbruch: NurExone Biologic, Regenxbio und Lineage Cell Therapeutics setzen auf unterschiedliche Zukunftstechnologien | ||
| OVID THERAPEUTICS | 2,020 | -1,94 % | Ovid Therapeutics Inc.: Ovid Therapeutics Appoints Dr. Petra Kaufmann as Chief Medical Officer | NEW YORK, Dec. 02, 2025 (GLOBE NEWSWIRE) -- Ovid Therapeutics Inc. (Nasdaq: OVID), a biopharmaceutical company developing small molecule medicines for brain disorders with significant unmet need,... ► Artikel lesen |
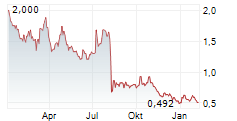
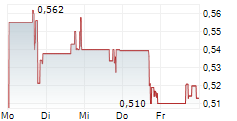
LONGEVERON INC gehört der Branche Biotechnologie an. Die relative Kursveränderung beträgt aktuell +2,07 % bei einem Aktienkurs von 0,721 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu LONGEVERON INC finden Sie auf Wallstreet Online.